Membrane carbohydrates are chemically bound to lipids resulting in glycolipids and to proteins to form glycoproteins. Other membrane carbohydrates are glycosaminoglycans as part of proteoglycans, which insert their amino acid chain among the lipid fatty acid chains (Figure 1). Although some carbohydrates can be found in intracellular membranes, most of them are located in the outer hemilayer of the plasma membrane, facing the extracellular space. The synthesis of membrane carbohydrates begins in the endoplasmic reticulum, but it is in the Golgi apparatus where they are modified and grown by adding many new monomers to form complex carbohydrate polymeres.
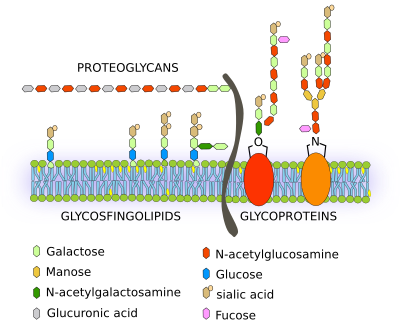
Three types of glycolipids are found in membranes: glycosphingolipids, which are the most abundant in the animal cells, glycoglycerolipids, and glycophosphatidylinositol. Glycoglycerolipids are more frequent in the plasma membrane of plant cells. However, most membrane carbohydrates are found linked to proteins, known as glycoproteins. Nearly all membrane proteins have carbohydrates, but only 5 % of lipids are glycolipids. Carbohydrates of the plasma membrane as a whole are referred to as glycocalyx. In some cell types, glycocalyx is so developed that it can be observed with the electron microscope. For example, in enterocytes, the glycocalyx can be extended more than 1 µm in length from the plasma surface. In this way, the cell is covered by a coat of carbohydrates that can be up to 2 to 10 % of the membrane weight. The glycocalyx development depends on the cell type.
Membrane carbohydrates are sites of cell recognition and adhesion, either cell-cell recognition or cell-pathogen interactions. For instance, blood groups are determined by cell surface carbohydrates of erythrocytes, and they also have the ability to trigger immunological responses. After an infection, endothelial cells near the injured tissue expose a type of proteins, known as selectins, in their plasma membranes. Selectins recognize and bind carbohydrates of the plasma membrane of lymphocytes that flow in the bloodstream. In this way, lymphocytes get attached to the blood vessel walls, can cross the endothelium and move to the infection focus. Carbohydrates as recognition molecules are important during embryo development. They also have a structural role as a physical barrier.
Sometimes, membrane carbohydrates are modified. For example, cancer cells show a different carbohydrate repertory, and a type of cancer cell can be identified by its distinct superficial carbohydrate set. These carbohydrates may modulate the ability of a cancer cell to receive and understand molecular information coming from other cells. In addition, a tandem repetition of sialic acid boosts malign features of cancer cells, such as proliferation, invasion, migration, adhesion and metastasis. GD3 ganglioside is found in melanomas, and it is a target for treatment.
Carbohydrates of the plasma membrane are major recognition and attaching sites for pathogens during infection. Virus, such as the influenza virus, pathogenic E. coli bacteria, and some protozoa, need to be attached to the cell surface before entering the cell, otherwise they will be swept by the cleaning mechanisms of the body. These pathogens have proteins, known as lectins, that bind to specific carbohydrates of particular cells. Thus, the type of cell to be infected depends on the carbohydrates they show in the plasma membrane. Vertebrates, invertebrates and protozoa bear different set of carbohydrates in their cells. Curiously, some pathogens are able to "dress" superficial carbohydrates similar to those of the host cells. In this way, they cannot be detected.
-
Bibliography ↷
-
Bibliography
Fuster MM, Esko JD. The sweet and sour of cancer: glycans as novel therapeutic targets. Nature reviews cancer. 2005. 5(7):526-542.
-
 Proteins
Proteins