There are several methods for fixating tissues that are selected according to the type of fixative, the structure to be fixed, and what we want to observe. Fixation methods can be grouped in two types: physical and chemical methods.
1. Physical methods
Physical fixation is accomplished by either a very quick freezing or by heating the samples at high temperatures. These procedures are used when chemical fixation affects the tissular structures we are interested in, when a very quick fixation is required, or when the techniques or the tissues need physical fixation.
Quick freezing is a suitable method for preserving molecular features of samples because they are not affected by any external chemical compound. Freezing must be very fast to prevent the formation of large ice crystals that may destroy the tissue structure. Thus, it is convenient to process sample sizes not larger than 2 mm so that freezing is homogeneous through all the tissue thickness, including the inner parts. A very fast freezing is achieved by plunge the samples in isopentane (-170 ºC) cooled in liquid nitrogen (-196 ºC), or by placing the sample in a metal sheet, which is partially immersed (not the sample) in liquid nitrogen, in dry ice and acetone (-70 ºC), or even in liquid helium (-268 ºC). If it is possible, before freezing, the samples should be pretreated and protected by a cryoprotectant substance for minimizing cellular damages. Cryoprotection is always a good practice, although it is not always possible. Dimethyl sulfoxide, glycerol, sucrose or mixed solutions containing some of these substances are the most common cryoprotectants. Once the freeze sample is sectioned, the tissue is melted and should be somehow protected.
Lyophilization, or freeze-drying, and cryosubstitution are techniques that remove the water (the ice) from frozen samples by sublimation, that is the solid water is transformed in gaseous water without going through the liquid water intermediate state. These techniques prevent chemical reactions happening in liquid aqueous environment, allowing a long-lasting preservation of the tissue. Cryosubstitution consists in the slow exchange of ice for a fixative solution. Thus, fixation is done in tissues that did not get degraded because they were frozen.
Fixation by heating is not common in histology because it introduces damages in the tissues. Heat coagulates proteins and dissolves lipids. However, it is a good fixative method for microorganisms because their morphology is preserved, and that helps in the identification. Nowadays, heating fixation is combined with chemical fixation. For example, samples are immersed in a chemical fixative and microwaved at 55 ºC. This temperature does not produce artifacts, increases the fixation speed, and decreases the fixation time from hours or days to minutes. Microwave is a good heater because temperature rises quite homogeneously and immediately through the sample. Heating baths are not recommended because they produce a heat gradient between the outer to the inner parts of the sample. It is thought that the increase in the fixation speed by microwave fixation is a consequence of the heat and not of the microwaves. Microwaves are sometimes used in later steps of the histological processing, particularly during staining.
2. Chemical methods
Chemical methods use aqueous solutions containing fixative substances that form bridges between tissular molecules and it results in the immobilization of the tissular compounds and prevents degradation of the tissue sample. To a greater or lesser extent, chemical fixatives affect tissues both chemically and physically. Retraction, distension, and hardening are common physical effects. There are two common methods for chemical fixation: immersion and perfusion. In any case, the fixative should get to all the regions of the sample as soon as possible.
Immersion
In the immersion method, tissue samples are plunged into the fixative solution (Figure 1). It is also used for blood smears or for fixing sections obtained from unfixed frozen samples. Some precautions should be kept in mind when fixing by immersion:

1) The piece of tissue should not be larger than 0.5 cm in thickness to allow the fixative enters the deepest part of the sample before the cells start to get deteriorated. The speed of the fixative for reaching the inner parts of the tissue depends on the type of fixative and the features of the tissue to be fixed. The size of the sample should be selected according to the speed of the fixative penetration. Samples not larger than 0.2 cm are recommended for slow diffusion fixatives. Tissue features need also to be considered. For example, fixation penetration is faster in loose tissues or samples with large spaces for diffusion.
2) The volume of the fixative is recommended to be 10 to 20 times larger than the volume of the sample.
3) Osmolarity of the sample and the fixative solution need to be as similar as possible.
4) The pH of the fixative solution should be close to the physiological pH of the sample.
5) For a similar type of sample, fixation time depends on fixative features: diffusion and fixation speed (intensity and speed of making bridges between proteins or how quick proteins coagulate). The fixation time must be enough for a proper fixation, but not too lomg because it may cause artifacts in the sample. A gentle agitation of the sample during fixation is recommended for increasing the surface fixative renewing, which reduces the fixation time. 24 hours of fixation is common for most of the fixatives. However, for some of them, like formaldehyde, fixation time may last for a week.
Perfusion
In this fixation method, the fixative solution is introduced through the vascular system and reaches all the cells of the tissue via the capillary net (Figure 2). It is possible to fix a whole animal if the fixative solution enters through the left ventricle of the heart. Then, the fixative solution is propelled by a peristaltic pump through the vascular branches of the arteries emerging from this ventricle. If we want the fixative in the lungs, the fixative solution is introduced through the right ventricle. By perfusion, a single organ can be fixed if the fixative solution enters by the main artery that irrigates the entire organ (Figure 3). Fixation by perfusion is not always possible, like in many biopsies or in plants.

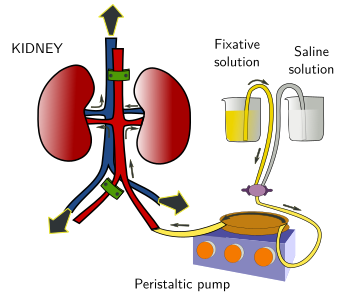
Fixation by perfusion is more effective than fixation by immersion because the fixative solution gets in contact with all cells of the perfused structure very quickly. In this way, the penetration speed of the fixative is not a limiting feature.
Before the introduction of the fixative in the vascular system, the blood should be removed with an oxygenated saline solution. Otherwise, the fixative may fix blood, coagulates it and produces thrombus. This may seal some parts of the vascular circuit that prevents the fixative solution entering some parts of the sample. As in the fixation by immersion, the pH and osmolarity of the fixative solution, and the fixation time, must be set properly.
Another parameter to keep in mind is the pressure of the fixative solution when entering the sample, which should be similar to the blood pressure in the living animal. The fixation pressure can be selected by using peristaltic pumps (Figures 2 and 3), and by gravity (rising or decreasing the height of the fixative container from the animal). This is important, because a low pressure may prevent the fixative solution to reach every capillary duct of the sample, but a high pressure may break blood vessels and the tissular structure.
 Fixation
Fixation 