Transcytosis is the vesicular transport of cargoes (molecules) between two plasma membrane domains of a cell. This mechanism was first posed by Palade (1950) for the transporting of molecules from one side to the other of the endothelial cells. He observed electron microscopy images suggesting the formation of vesicles in the plasma membrane domain facing the blood, crossing the endothelial cytoplasm and fusing with the plasma membrane facing the basal lamina and connective tissue. Epithelia are layers of cells separating two different environments, such as endothelium in blood vessels, lung epithelium or intestine epithelium. These cells have two plasma membrane domains: apical y basolateral. That is why they are called polarized cells. Epithelial cell need to keep the molecular identity of these two domains, but at the same time they need to communicate between each other. Part of this communication is by transcytosis. Although transcytosis is a mechanism present in most epithelial cells, it can be found in other cell types as well, such as neurons and osteoclasts. Transcytosis prevent molecules enclosed in vesicles by endocytosis ending up in lysosomes, so they skip degradation. A number of molecules are transported by transcytosis: immunoglobulins, insulin, quimocine receptors, lipoproteins, DNA fragments, enzymes, some viruses, some toxins, etcetera.
Endothelial cells move a large amount of molecules between the blood serum and the surrounding tissues by transcytosis. Plasma soluble molecules are usually included in vesicles by endocytosis, without needing specific receptors. The vesicle goes from one plasma membrane domain to the other, that is, there is no fusion with endosomes. This transport is fast (about 30 seconds) but not very specific. However, it is not totally random and some selection is done based on the net negative electric charge of the molecules. In this way, immunoglobulins, low-density lipoproteins, micronutrients, iron, B12 vitamin, and other molecules are transported. Only a few molecules, such as albumin and orosomucoid proteins, are specifically caught by receptors.
Entherocytes, cells of the intestine epithelium, are columnar cells showing transcytosis. Unlike endothelial cells, where apical and laterobasal membranes are very close, transcytosis in entherocytes is a rather long path and needs the cytokeleton and endosomes as intermediate organelles. Entherocyte transcytosis usually begins with clathrin coated vesicles that specifically capture cargoes (receptor mediated endocytosis). These vesicles fuse with early endosomes. There are two populations of primary endosomes: basolateral and apical (Figure 1), each of them receiving vesicles from the closer plasma membrane domain. From early endosomes, vesicles are shipped to another type of endosomes found at the perinucler region, which are known as common endosomes. These endosomes receive vesicles from both apical and basolateral endosomes, and at the same time they selectively ship vesicles to both basolateral and apical plasma membrane domains.
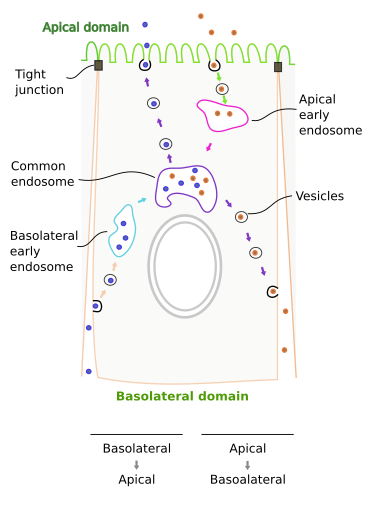
Two transcytosis pathways have been described in entherocytes: apical to basolateral and basolateral to apical. Immunoglobulins A (IgA) are antibodies released by plasmatic cells in the internal tissues of the body, but their functions are performed in the surfaces of the body. Thus, they have to be transported to epithelia and then cross intracellularly epithelial cells from basolateral domain to apical domain (body surface). Many plasmatic cells are found in the intestine mucosa, where they release IgAs. Epithelial cells synthesize receptors for IgA. The receptors are first synthesized in the endoplasmic reticulum, transported to the Golgi apparatus, where they are included in vesicles targeted to the basolateral domain of the entherocyte. Once the receptors are in the basolateral membrane, they recognize and bind IgAs, which causes oligomerization of IgA-receptors complexes. These molecular complexes (many IgA-receptors gathered together) are then endocytosed and the formed vesicles fuse with basolateral endosomes. IgA-receptors are again included in the membrane of new vesicles and shipped to the common endosomes. From common endosomes, new vesicles are formed with IgA-receptors in their membranes and are targeted to the apical plasma membrane domain. In the apical surface of entherocytes, receptors are hydrolyzed and IgAs are released (actually with a part of the receptor molecule atached). The receptor has a 100 amino acid sequence as a label, that is recognized by the trafficking machinery of the cell, and make possible to move IgA-receptor through the different compartments along the transcytosis pathway. This mechanism is also found in the epithelia of kidneys, trachea, liver and mammary glands. Lung respiratory epithelia are also capable of transporting IgA toward their free surface, that is, the lumen of the respiratory airways, by transcytosis. Although in this case IgA is released by different pH values between the surface and the inner tissues.
Curiously enough, transcytosis for immunoglobulins may happen also in the opposite direction: from apical domain to basolateral domain. For example, maternal immunoglobulin IgG travel from placenta to the fetus, or from milk to the blood vessels of the intestine. IgG are included in clathrin coated vesicles after being recognized by a receptor. The crystallizable part of the immunoglobulin is recognized by the receptor. Vesicles are then targeted to early apical endosomes, IgGs are later transported to the common endosomes, where they are included in vesicles that fuse with the basolateral domain (figure 1), and IgGs are then released into the interstitial tissues.
Transcytosis is not always for transporting extracellular molecules. It is sometimes for moving plasma membrane molecules from one domain to the other. Entherocytes and hepatocytes synthesize transmembrane proteins that are initially located in basolateral membranes, and are later transported to the apical domain by transcytosis. Some sphingolipids may travel in the opposite direction, apical to basolateral, by endocytosis too. Cholera toxin uses this movement of sphingolipids to cross the epithelium and avoiding lysosomes, since the toxin can bind sphingolipids. It is not well understood how sphingolipids are specifically selected in each endosome, but it may depend on the formation of lipid rafts and on the length and saturation level of their fatty acid chains.
Bibliography
Fung K, Fairn GD, Lee WL. 2018. Transcellular vesicular transport in epithelial and endothelial cells: Challenges and opportunities. Traffic. 19: 5-18.
García-Castillo MD, Chinnapen DJ-F, Lencer WI. 2017. Membrane transport across polarized epithelia. Cold Spring Harbour perspectives in biology. 9:a027912.
Tuma PL, Hubbard AL. 2003. Transcytosis: crossing cellular barriers. Physiological reviews. 83: 871-932.