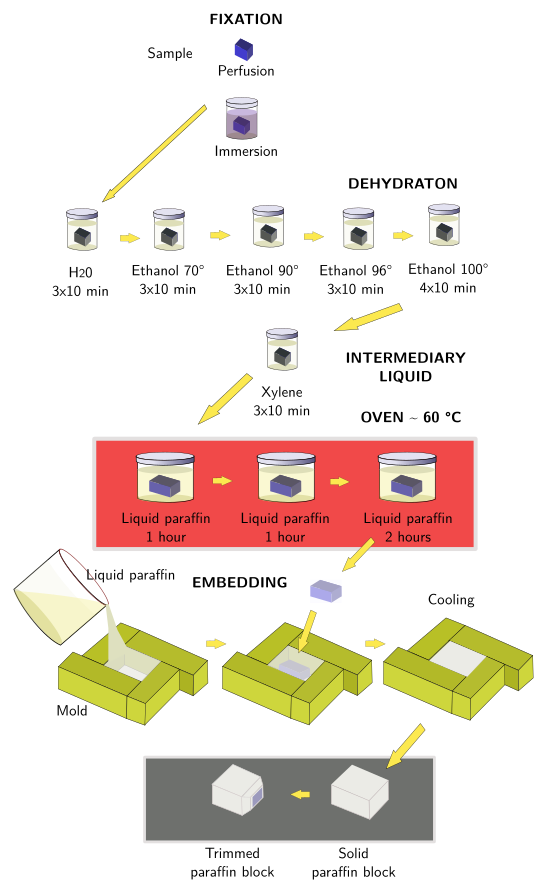
To study tissues at light microscopy, paraffin is the most widely used embedding compound. The paraffin embedding procedure of fixed samples is described below.
Paraffin is a wax-like substance composed of a mix of saturated hydrocarbons. It is solid at room temperature. The melting point is between 40 °C and 70 °C, depending on the composition of the hydrocarbons mix. Thus, hard paraffin at room temperature melts at higher temperatures, whereas soft paraffin melts at lower temperatures. Tough samples are recommended to be embedded in hard paraffin. Most used paraffin has a melting point close to 60 °C. Some substances can be added for modifying hardness, viscosity and fragility of paraffin.
Paraffin is not miscible with water, but tissues are mostly water. In addition, most fixatives are aqueous solutions. This means that water must be removed from tissues before they are infiltrated with liquid paraffin. It is done by dehydration of tissues with alcohol, commonly ethanol, in a graded series of alcohols startining in 50º and finishing in 100º (absolute o pure alcohol). All water needs to be removed for a good embedding. After dehydration, samples are transferred to an intermediary liquid, like xylene, benzene, propylene oxide, or toluene, which are miscible with both absolute alcohol and paraffin. These are clearing substances, and we can check their infiltration in the sample by observing how translucent the sample is. The immersion of the sample in the intermediary liquid is recommended not to be very long because the samples are hardened and getting sections might be more difficult.
The last step of the embedding procedure is to plunge the sample in melted paraffin. It is done in a stove at a temperature properly set for the paraffin type we are working with. For a complete replacement of the intermediary liquid with paraffin, three changes in fresh paraffin are recommended. How long the samples are incubated in paraffin depends on the intermediary liquid, size of the sample, and type of paraffin. For example, it should be longer for large samples. However, if the immersion last very long, paraffin may harden the sample. On the other hand, short incubation times may render poor paraffin infiltration, which can be worse than long incubation times. After the complete substitution of the intermediary liquid by paraffin, the sample is placed properly oriented in a metallic mold filled with liquid paraffin. Then the mold with the infiltrated sample and the paraffin surrounding the sample are left at room temperature or onto a cooled plate so that paraffin gets solid.
Common steps for paraffin embedding (Figure 1):
 Embedding
Embedding