Mastocytes are often found in the connective tissue proper. They differentiated from myeloid cells and contain many granules with substances such as histamine and heparin. Masctocytes are rounded cells with metachromatic granules (showing a different color from the color of the dye). Mastocytes are related are involved in immune responses, particularly with allergic and hypersensitivity responses.
Mastocytes were first described by Paul Ehrlich at the end of the 1800s as cells that could be stained with aniline dyes and showing metrachromatic granules in the cytoplasm. He named them "Mastzellen" (well-fed, in German), because he thought that the granules were food leftovers after phagocytosis and digestion. Food was incorporated from the surrounding tissue. Mastocytes have been found in many vertebrate species studied ever since, fish included. That is why they probably are ancient phylogenetic cells.
1. Morphology
At light microscopy, mastocytes are round or ovoid. In humans, they are 8-20 µm in size, depending on the organ where they are found. The nucleus is not lobulated and occupies a central position. The more salient feature is the large amount of metachromatic granules in the cytoplasm. That is, they are able to change the color of the staining dye. Metachromasia is clearly visible when they are stained with toluidin blue because granules show a reddish color (Figures 1 and 2). This is mostly caused by heparin, a sulfated glycosaminoglycan.
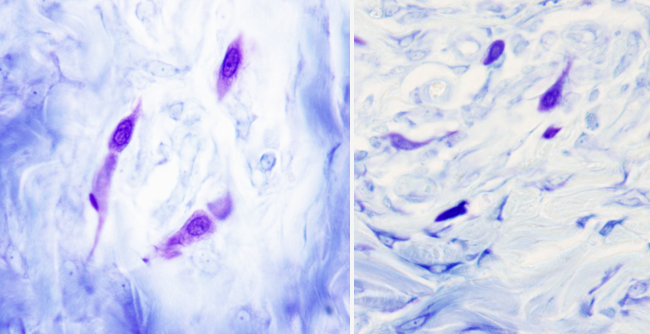

Microvilli and small folds are visualized at the cell surface with the electron microscope. Mastocytes do not have many mitochondria, endoplasmic reticulum profiles are short, and show many free ribosomes. The secretory granules are about 1.5 µm in diameter, are membrane-bound organelles, and the granule content appearance varies according to the stored substances and to the animal species. For example, in rodents, granule content looks like fine granulated, whereas in humans the granule content may be organized in thin and concentric sheets. The appearance of the granule content may sometimes be related to its function. For example, granules that contain chymase enzyme show crystalline or homogeneous appearance in humans. At electron microscopy, different densities are observed in the granules of the same mastocyte, which means a different content in the same cell.
Besides granules, there are non membrane-bound structures in the cytoplasm known as lipid bodies. They are more frequent in humans than in rodents, and are places for storing arachidonic acid.
Mastocytes of an individual do not constitute a homogeneous population. For example, electron microscopy studies show that different types of mastocytes contain specific sets of granules. Two major types of mastocytes have been identified based on morphological and functional features: the connective tissue mastocytes (Figures 3) , which are found in the connective tissue of the skin, peritoneum and serous layers of many organs, and mastocytes of the mucosa, which are found in the intestinal and respiratory mucosas (Figure 4). Connective tissue mastocytes show red granules when stained with safranin, probably because of the high concentration of heparin, whereas mastocytes of the mucosa lack this type of granules. However, detailed functional studies suggest more than two populations. For example, mastocytes in the same tissue, but located in different areas, show distinct sets of proteases in their granules. It seems that these features appear once the mastocytes arrive at the places where they have to perform their functions.
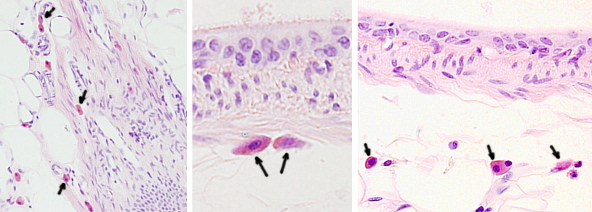
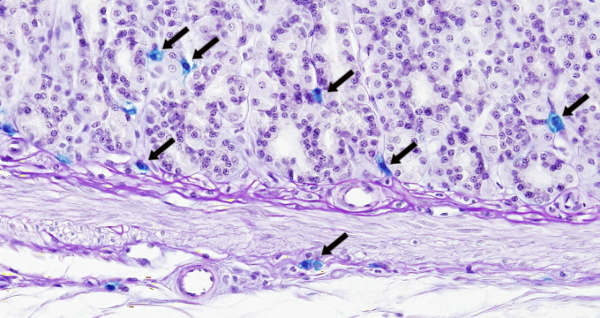
An interesting classification of mastocytes is based on their proteases content, because the cellular functions rely on the proteases profile that mastocytes put to work. Mastocytes from different locations have different protease profiles. Human mastocytes can be divided into those containing triptases (located in the respiratory and digestive mucosa, close to T lymphocytes), and those containing triptases and chimases, together with other enzymes (found in the epidermis, intestine and stomach submucosa, myocardium, lymphatic nodules, conjunctive and synovium). Later, another type of mastocytes was discovered containing chimase without triptase (found in the intestine and stomach submucosa). However, regarding other features, such as how they respond to T lymphocytes, more subtypes of mastocytes can be characterized. Altogether, it can be said that mastocytes form a heterogeneous population.
2. Origin and distribution
Since mastocytes show similar functions and morphology than some leukocytes, it was initially thought that mastocytes differentiate from leukocytes or from a common myeloid progenitor. In 1977, it was observed that mastocytes develop from a hematopoietic precursor cell located in the bone marrow. In addition, and unlike basophils, mastocytes abandon the bone marrow as undifferentiated agranular cells and are transported through the blood stream as undifferentiated cell (morphologically, they are agranular leukocytes). Therefore, there is a circulating population of progenitor cells in the blood, but they are also found in the lymphatic ganglia and other tissues. These progenitor cells can not be morphologically distinguished. After a while in the bloodstream, undifferentiated mastocytes exit blood vessels and populate connective tissues to complete differentiation and become mature functional cells. This last process is induced by tissue local signals, mostly coming from fibroblasts, endothelial cells and reticular cells. Not all mastocytes differentiate after leaving the blood stream, but some of them remain as a pool of undifferentiated cells.
The ability of mastocytes to populate, move and interact with the extracellular matrix of different tissues relies on their adhesion capabilities. Adhesion mostly relies on integrins, and in extracellular matrix proteins such as laminins, fibronectins and vitronectins. In humans, α4-β1 integrin is important. Other integrins are also relevant, for example, those mastocytes expressing the β7 subunit are frequently found in the mucosa. Mastocytes can also be adhered to other cells like epithelial cells, fibroblasts and lymphocytes. However, these cell-cell adhesions seem to be related to cell communication. The number of mastocytes in a tissue is relatively constant during physiological conditions.
Mastocytes are ubiquitous and can be found in almost any tissue. Unlike other cells with hematopoietic origin, mature differentiated mastocytes populate non-pathological tissues. Mastocytes can move and are concentrated around small blood and lymphatic vessels, and nerve fascicles. They are more abundant in the connective tissues, such as the dermis, in the urinary tracts, and in the respiratory and digestive ducts (in the connective tissue of mucosa and submucosa). It means that mastocytes are frequently found in those structures where the external and internal environment are close, and are potential entry pathways for pathogens. Mastocytes also live in vulnerable regions like joints and peritoneum. They are not present in the bone matrix, cartilage, nor in the cornea, all of them non-vascularized tissues. In the human skin, mastocytes are concentrated close to the epidermis and are more abundant in the skin of the nose and cheeks than in the abdominal skin. In humans, there is mastocyte reservoirs in the connective tissue surrounding the hair follicles. The uterus is another organ with many mastocytes, where the number and granule content is regulated by reproductive hormones. Many mastocyte progenitors are found in the adipose tissue. In the nervous tissue, mastocytes are mostly associated with the leptomeninge, but they can also be found in the hypothalamus, thalamus and spinal cord duramadre meninge. Since there is a constant incoming of new mastocytes into the tissues, it is thought that the total number is regulated by apoptosis induced by signal coming from the surrounding tissue. In humans, it has been estimated that the total population of mastocytes might form an organ similar in size to the spleen.
3. Functions
Mastocytes have to be activated to perform their functions. This may be done by activating the Fc-IR plasma membrane receptor, which recognizes the constant domain of E immunoglobulins. This receptor is also found in basophils, Langerhans cells and monocytes. Other non-immune molecules can also activate mastocytes, such as neuropeptides, some basic substances, some drugs like opioids, and some others. Mastocytes harbor a broad variety of receptors in the plasma membrane that allows them to respond to a great diversity of stimuli.
Mastocytes are probably among the first cells activated during inflammation. Once activated, mastocytes release the granule content by a mechanism known as degranulation. There are two types of degranulation: explosive (anaphylactic or mixed exocytosis) and slow. During explosive degranulation, the granules fuse to one another and then fuse with the plasma membrane, resulting in a massive and instant content release. The fusion process may form channels between adjoining granules that allow the release of the content from those granules located deeper in the cell. Explosive degranulation is triggered by the gathering of FCεRI receptors after the binding and crosslinking of IgE immunoglobulins with di- or trivalent antigens. During this process, granules swell and decrease in density. Large membrane-bound compartments are formed in the peripheral regions of the cytoplasm before they are fused with the plasma membrane. Thus, this mechanism leads to secreting large amount of substances in a very short period of time. It has been observed that these secretory compartments are already present 3 minutes after the stimulation. Strikingly, it is very rare to observe these compartments in vivo and most descriptions of this type of degranulation comes from cell culture. Explosive degranulation is observed during allergic responses. Slow degranulation releases small amounts of granule content for a longer time. This mechanism is more frequent, and it happens in infiltrated tissues, such as some tumors and chronic inflamed tissues. Slow release was proposed after the observation of mastocytes with cytoplasmic regions containing empty granules not connected between each other nor with the plasma membrane. These mastocytes also contain more endoplasmic reticulum and more abundant vesicles, which were sometimes fused with granules. This is the most frequent morphology of activated mastocytes in tissues with chronic inflammation. The cellular mechanism for the release of the granule content depends on the vesicular trafficking. In fact, small amounts of granule content are included in small vesicles that pinch off from the granules themselves. Then, they are moved toward the plasma membrane and exocytosed. A similar number of endocytosis vesicles fuse with granules to keep the granule membrane constant. Explosive degranulation is massive, non-accommodating and non-selective regarding the granule type and granule content. Slow degranulation is long-lasting, may change the release flow intensity, and may select the granule type and granule content to be released.
Mastocyte performs many functions, which are sometimes similar to those of other immune cells, such as basophils, monocytes and neutrophils. Mastocytes are able to do phagocytosis, antigen processing, release of cytokines and vasoactive substances. Allergic responses largely relies on mastocytes.
In the 1950s, some studies had already been demonstrated a positive correlation between the amount of histamine that a tissue can release and the number of mastocytes in that tissue. Histamine was thought to be involved in allergic and anaphylactic processes, so that mastocytes could contain histamine and participate in these processes. Later, it was observed that basophil also contain histamine, and therefore they would be involved in these functions too.
Cytoplasmic granules, besides enzymes, contain molecules known as mediators, which influence the activity of other cells. Mediators can be divided in preformed mediators (primary mediators) and precursors (secondary mediators). Primary mediators are histamine, heparin, and chemotactic molecules for eosinophils and neutrophils. Precursors, such as prostaglandins and leukotrienes are derived from lipids and from arachidonic acid, and other precursors non-derivated from lipids are interleukins (3, 4, 5, and 6), platelets activator and necrosis activators.
Allergy. Although many cells are involved in allergy responses, mastocytes are the cells that trigger the process (Figure 5). It begins when the receptors Fc-IR bind immunoglobulin E (IgE)-antigen complexes. IgE is released by other cells after the antigen (allergen) is recognized. The first mastocyte response is the fast degranulation (in minutes) that releases the granule content with pro-inflammatory molecules. In many allergic responses, there is a second phase starting at about 2 to 4 hours later, which releases cytokines and chemokines. The global effect of the allergic response depends on the tissue where it is taking place.
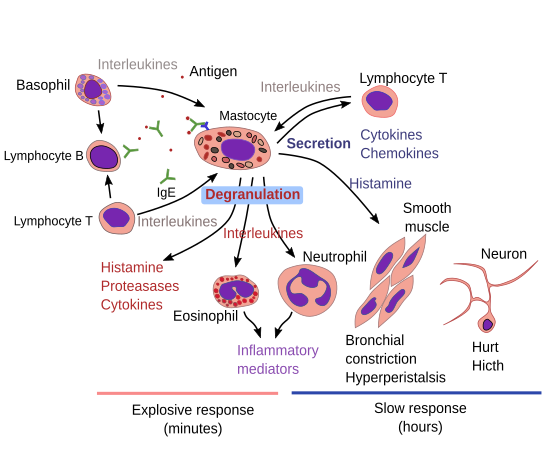
Allergic reactions may be local, as in nasal mucosa during rhinitis and as in lungs during asthma, or may be general when an anaphylactic shock is going on. Generally, the release of histamine and other lipidic mediators increases the permeability of blood vessel walls, facilitating the exit of plasma molecules to the surrounding tissues, which ends up as edemas. Leukotrienes induce the contraction of the smooth muscle layer in the respiratory ducts. In addition, mastocytes release chemotactic factors to attract eosinophils and neutrophils, which partially counteract the reaction by releasing anti-histamine by eosinophils and defense activity molecules from neutrophils. The symptoms of allergic reactions are pruritus, skin edemas, cutaneous erythema (redness of the skin), swelling of the nasal mucosa and aqueous nasal secretion, spams and increase of the mucous secretion by respiratory ducts.
The strategic position of mastocytes around blood capillaries and nerves, and the high amount of chemical substances in their granules, suggest that they are also involved in inflammatory and immune responses. Many receptors have been found in the plasma membrane of mastocytes and many chemical mediators in their granules. Mastocytes may participate in the response mechanisms to more than 20 non-allergic pathologies, such as arteriosclerosis, atopic dermatitis, cystitis, migraines, osteoporosis, psoriasis, and several tumors.
-
Bibliography ↷
-
Bischoff SC. 2007. Role of mast cells in allergic and non-allergic immune responses: comparison of human and murine data.. Nature reviews in inmunology 7: 93-104.
Metcalfe DD, Baram D, Mekori YA. 1997. Mast cells. Physiological reviews 77:1033-1079.
Molderings GJ. 2010. Mast cell function in physiology and pathophysiology. Biotrend reviews 5:1-12.
Sobota W. 2008. Histología (2ª ED.) Ed. Panamericana. ISBN: 9788498351781.
-