Fibroblast is the characteristic and most abundant cell type of the connective proper tissue. The fibroblast main function is to synthesize and maintain the extracellular matrix of tissues. Although the suffix "blast" is generally used for undifferentiated cells, and the suffix "cyte" for differentiated cells, fibroblasts are actually differentiated cells, but showing a high proliferation capability and a distinct metabolism. Thus, fibroblast and fibrocyte are two physiological states of the same cell type, being fibroblast the most active and fibrocyte the more quiet. Many authors suggest that fibroblasts are young metabolically active and proliferating cells, whereas the term fibrocyte should be used for old and low-rate secretory cells. However, the term fibroblast is commonly applied to both cell types.
Currently, fibroblast is a thoroughly studied cell type in tissue regeneration research and it has been frequently selected for cell culture experiments since long time ago. In 2006, embryo and adult mouse fibroblasts were transformed into cells showing embryo stem cell features by using retrovirus that modified only 4 transcription factor genes. These transformed cells were named IPSc (Induced Pluripotent Stem Cells). In 2007, IPSc were obtained from human fibroblasts. These breakthroughs opened new research pathways for tissular regeneration, at the same time avoiding the use of embryos.
1. Morphology
Fibroblasts show a variety of morphologies, both in shape and size, depending on the organ they are found and on their cellular activity level. Generally, they are elongated (Figures 1 and 2) or star-like cells, with cytoplasmic projections that can be short and wide, or long, thin and branched. These last cell protrusions allow physical contact between close fibroblasts. Gap junctions can be found in these contacts. They can also physically contact other cells, such as neurons, muscle cells, endothelial cells, leukocytes, and others. Fibroblasts also communicate with other cells by using the extracellular matrix as an intermediate and by releasing molecules.
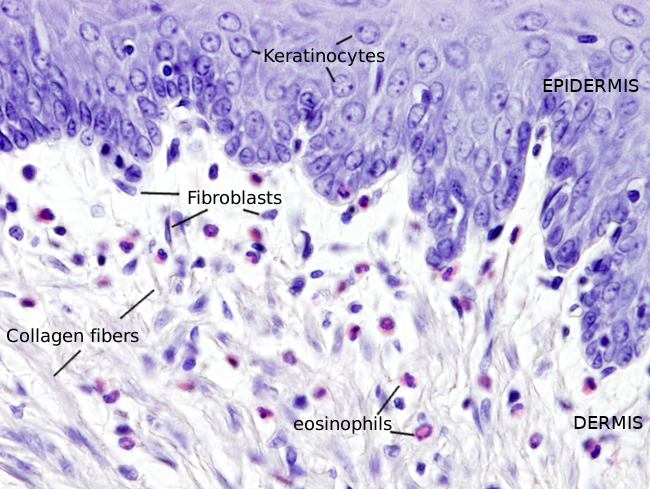
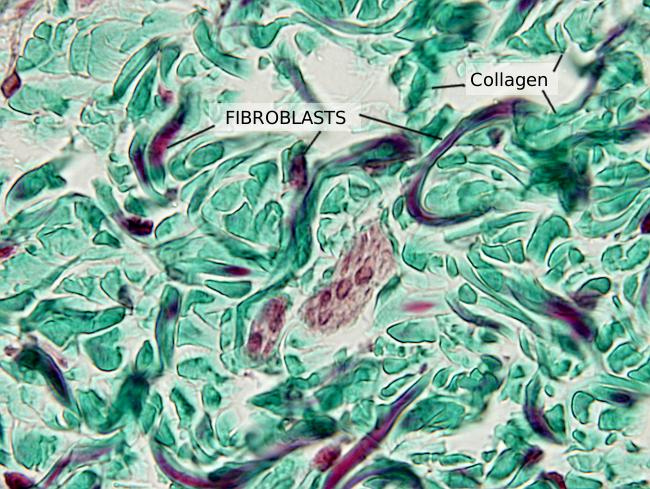
Fibroblasts usually show one elongated nucleus and scarce cytoplasm. They commonly have a well-visible nucleolus, whereas fibrocytes do not, which may be a landmark to distinguish both cell stages. At transmission electron microscopy (Figures 3 and 4), fibroblasts do not usually show well-developed organelles. However, active fibroblasts are larger, with more cytoplasm and abundant organelles (Golgi apparatus, endoplasmic reticulum and ribosomes) involved in extracellular matrix synthesis.
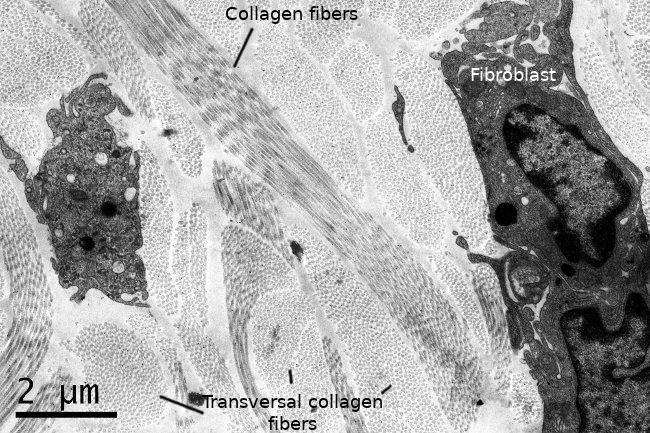
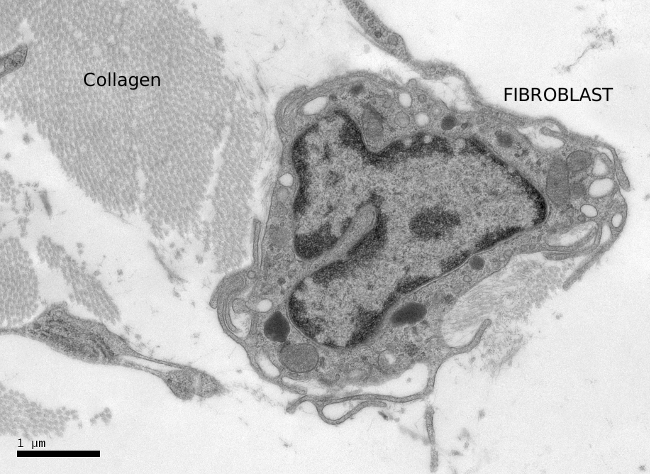
Fibroblasts show a well-differentiated cytoskeleton. Actina and alpha-actinin are found in the cell periphery, where myosin is also present. Alpha-actinin makes possible the anchoring of actin filaments to the plasma membrane through integrin transmembrane proteins. Integrins connect the cytoskeleton with collagen and fibronectins of the extracellular matrix. It is a weak binding that can be formed and removed easily. All these structural components allow fibroblasts to move at around 1 µm/min speed through the extracellular matrix.
2. Origin, distribution and diversity
The morphological diversity of fibroblasts is determined by the embryonic origin and the environment where they develop. Fibroblasts are characterized by the expression of mesenchymal cellular markers like vimentin, and type I collagen. Furthermore, they lack molecular markers found in other cell lineages. During embryo development, fibroblasts may be generated from ectoderm (neural crests) and mesoderm. However, fibroblasts with the same embryonic origin form heterogeneous populations.
The most deeply studied fibroblasts are probably those found in the dermis. The gene expression pattern of dermal fibroblasts is different from fibroblasts of other parts of the body. Gene expression profiles appear to be inherited as a result of their embryonic origin, remarkably influenced by Hox genes, that provide positional information. Genes expressed in fibroblasts have been related with growth, differentiation, cell migration, extracellular matrix production and lipidic metabolism.
Two types of heterogeneity can be distinguished in dermal fibroblasts. a) Fibroblasts form different populations according to the body location: rostral or caudal, ventral or dorsal. The position is important for fibroblast function, and they somehow know the spatial location. For example, fibroblasts are thought to be involved in the induction of hair follicles, sweat glands, and feathers in birds. All these structures show different distribution, density and features in different positions of the body. Furthermore, fibroblasts in different places are derived from particular embryonary structures. Face dermal fibroblast develop from neural crests (ectoderm), back dermal fibroblasts from the somitic mesoderm, ventral dermal fibroblasts from lateral mesoderm. Even coming from the same embryonic source, the final position influences fibroblast functioning, dermal regionalization and fibroblast morphology. b) There is local heterogeneity as well. Dermis is divided in an upper papillary region and deeper reticular region. Papillary fibroblasts are different from reticular fibroblasts in physiology and regarding the extracellular matrix molecules they synthesize and release. In papillary dermis there are thinner and scattered collagen fibers, more type I and III collagens and more decorin than in the reticular dermis. Papillary fibroblasts are more affected by age than reticular fibroblasts. In addition, fibroblasts associated to hair follicles form a third population that in turn can be divided in two subpopulations: those in the interior of the hair follicle that contribute to the bulb size, follicle length and diameter, and those surrounding the basal membrane of the follicle epithelium.
3. Functions
Fibroblasts perform many functions. The most common one is synthesizing and maintaining many components of the extracellular matrix, such as collagen, and reticular and elastic fibers, as well as ground substance, including glycosaminoglycans, proteoglycans and glycoproteins. Fibroblasts contribute to the skin homeostasis in adults. This function is done by two fibroblast populations with different roles. They are also involved in tissular structural organization. For instance, fibroblasts cooperate in the formation of the skin during embryo development. They are essential during wound healing by forming groups of fibroblasts at the edges of the wounds and releasing extracellular matrix to form scars. They also appear to be involved in other less known functions. For example, fibroblasts participate in the first stages of the immune response.
The fibroblast plasticity makes this cell type easily cultured in vitro, and therefore very interesting for research purposes. Fibroblast culture is extensively used in cosmetic industry. For example, face wrinkles are a consequence of collagen lost. Collagen, elastin and proteoglycans are more slowly synthesized during aging. Cultured fibroblasts provide those molecules that can be injected in the skin. A recent innovation is the injection of fibroblasts in the skin. For that, fibroblasts are obtained from a skin biopsy, usually from the region behind the ears, the cells are grown in vitro, and then these fibroblasts are injected in the region to be filled in the same individual.
-
Bibliography ↷
-
Sriram G, Bigliardi PL, Bigliardi-Qi M. 2015. Fibroblast heterogeneity and its implications for engineering organotypic skin models in vitro. European journal of cell biology. 94: 483-512.
-