1. Morphology
2. Number
3. Functions
- Neuronal activity
- Glia limitans
- Blood–brain barrier
- Blood circulation
4. Pathologies
5. Others
Astrocytes are cells found in the central nervous system: encephalon and spinal cord. Together with oligodendrocytes, microglia, and Schwann cells, astrocytes are glial cells. They are the more abundant type of glial cell. For a long time, astrocytes have been regarded as minor cells in the nervous system when compared with neurons. Astrocyte function appeared to be limited as housekeepers of neurons and to form barriers in the borders of the central nervous system. However, many and varied functions are ascribed to astrocytes nowadays, such as control the blood-brain barrier, regulation of blood circulation, neuron metabolic support, antioxidant role, neurotransmitter recycling, regulation of synpatogenesis and synaptic function. Some functions show up only during pathological processes or during development. Astrocytes have are also involved in modulating the information processed by neurons. It is interesting that during the primate evolution, the ratio of astrocytes/neurons has increased in the encephalon.
1. Morphology
Glial fibrillary acidic protein (GFAP) is a protein specifically found in the cytoskeleton of astrocytes. The name astrocyte is because of the star-like morphology of these cytoskeletal intermediate filaments. However, the astrocyte cell morphology may be variable. For example, those locate in the gray matter are known as protoplasmic astrocytes and those in the white matter as fibrous astrocytes. There are also highly specialized astrocytes, such as the Bergmann glia in the cerebellum and Müller glia in the retina (Figure 1).

Protoplasmic astrocytes have cell expansions that may branch several times. The terminal ends of these processes are called end-feets, which line blood vessels, pial surfaces and wrap synapses (one astrocyte may wrap thousands of synapses). Fibrous astrocytes are found in the white matter and show thinner cell expansions with few branches. End-feet of fibrous astrocytes wrap Ranvier nodes and blood vessels in the axonal tracts.
2. Number
Astrocytes are the most abundant glial type in the brain (Figure 2). The number of astrocytes is variable depending on the brain area, but they are about 20 to 25 % of the volume of many brain areas. In cat and rat brains, astrocytes are as numerous as neurons.
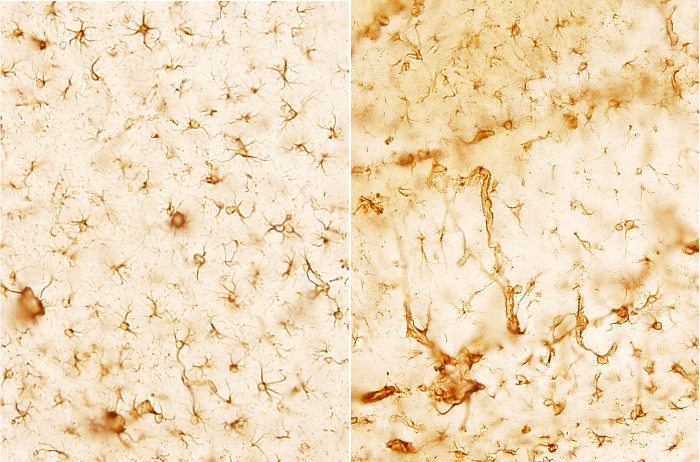
Astrocytes are mostly generated during the perinatal period, once the massive production of neurons is almost over. This has been found in humans and cats. During this period, astrocytes may be differentiated from several sources. For example, in the cerebral cortex, astrocytes can differentiate from the radial glia, ventricle walls, and from NG2 undifferentiated glia. Initially, all these cells become progenitor astrocytes, which differentiate directly into astrocytes, or go first through a number of divisions to increase the future local population of mature astrocytes. Astrocytes may also proliferate in adult tissues. Local production of new mature astrocytes in the adult period may come from the mitotic activity of some progenitors that remain inactive and undifferentiated for a long time. During the perinatal period of cats, the ratio glia/neuron is 0.86, whereas in adults is 1.4. It means that astrocytes proliferate in a way that outnumber neurons.
3. Functions
Astrocytes have been regarded as a functionally homogeneous population, but it is not. There is a diverse variety of functions performed by astrocytes that relies on in the expression of a particular set of proteins, such as plasma membrane ion channels or other membrane proteins. They lead to different populations of astrocytes to respond to similar stimuli in different ways.
Neuronal activity
Compared to neurons, a rather passive function has usually been assigned to astrocytes. They were thought to be involved in neuron homeostasis, feeding neurons, mechanical support and contribute to the normal function of the nervous tissue by isolating synapses (preventing interference between close synapses). It was found that astrocyte also clear or inactivate extracellular neurotransmitters like glutamate, and remove and redistribute extracellular ions such as potassium, which above some levels is dangerous for neurons. All these functions contribute to a proper function of synapes. However, it is currently known that astrocytes perform a much more active role in neural transmission. The end-feets of astrocytes are around neurons, synapses and Ranvier nodes.
Astrocytes respond to a number of neurotransmitters, such as monoamines, neuropeptides, GABA, acetylcholine, nitric oxide and endocannabinoids. There are receptors in the plasma membrane of astrocytes for these neurotransmitters. Astrocytes are no activated synaptically, but through the so-called volume transmission. Therefore, astrocytes may respond to neuronal activity. Astrocytes around an active synapses are able to respond to neuronal activity by rising the inner calcium concentration. Once activated, astrocytes can release molecules known as gliotransmitters, such as glutamate, ATP, and adenosine. Receptors for gliotransmitters can be found in neurons, and their activity can be therefore modulated. Currently, the model of the synapse is the tripartite synapse (Figure 3), which includes presynaptic element, postsinaptic element, and the glia that wraps the two of them.
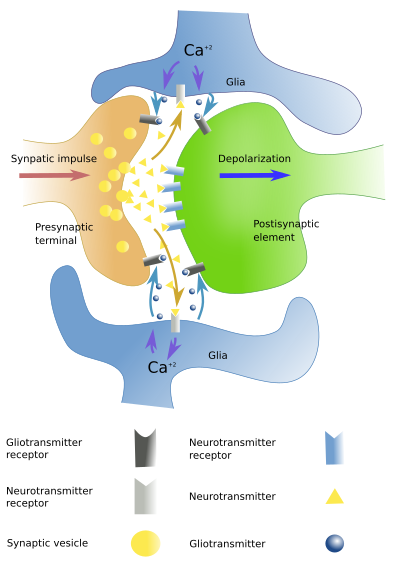
Astrocytes influence neurons in other ways. They are involved in the formation, maturation and maintaining of synapses. They can also remove synapses by phagocytosis. During development, astrocytes help to establish neuronal connections and facilitate the axonal migration.
Tthere are other examples of functional interactions between neurons and astrocytes. In some cortical areas and in the striatum, supopulations of astrocytes are activated only if a particular set of neurons are working. It means that astrocytes may specifically respond to some kind of neuronal processing. In addition, astrocytes may influence some types of neurons, but no others. The rhythm pattern of some neuronal pacemakers need healthy astrocyte networks. In the hypothalamus, thanks to the presence of receptors for leptin, astrocytes are able to modulate the food intake circuits. In addition, insulin activates hypothalamic astrocytes. If these astrocyte receptors for insulin are removed, there are alterations in the food intake and in the control of glucose levels.
Glia limitans
Astrocytes line the outer surface of the brain and spinal cord (Figure 4). Astrocyte end-feet (monkeys) or whole astrocytes (mice) form a sheath that isolate neurons from meningeal membranes that cover the central nervous system. This outer layer made of astrocytes is called glia limitans. Astrocytes of glia limitans are anchored to a basal lamina, and the basal lamina to the inner pial membrane. The function of glia limitans is unknown, apart from being a physical layer that keeps neurons separated from other non-nervous elements. In mice, the glia limitans is made up astrocytes bodies that cover the central nervous system surface, except the olfactory bulb and cerebellum. Some authors suggest that astrocytes involved in forming glia limitans are different from protoplasmic and fibrous astrocytes.

Blood–brain barrier
Paul Hehrlich noticed that the encephalon and spinal cord were special structures because when he injected ink in the blood system all organs got stained excepting the central nervous system. There was something that prevented ink to get out from blood vessels in these nervous structures. This is because of the so-called blood-brain barrier, a structure made up of endothelium, pericytes, basal lamina and end-feet of astrocytes (Figures 5 and 6). In the central nervous system, endothelial cells are very close to each other, more than in any other organ. They have many tight junctions that seal the endothelial intercellular space. Thus, molecules that want to cross the endothelium need to cross through the endothelial cells. It looks like that the end-feet of astrocytes, which do not form a real barrier, may regulate these cell junctions and influence the molecular trafficking across the endothelial layer.
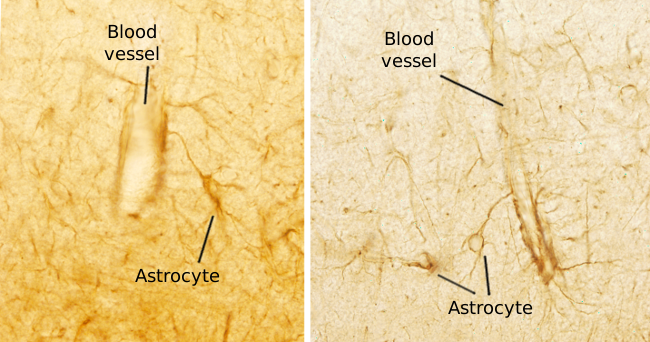

The term barrier is somehow misleading because it is not a strict barrier. There is an intense and selective exchange of molecules between the blood and the extracellular matrix of the nervous tissue. Even lymphocytes can cross it sometimes. Blood-brain barrier can be crossed by molecules with molecular weight under 500 kDa, liposoluble molecules and those that have a specific transporter. This selectivity may be a hurdle for some medicament to reach the neurons. It is curious that when some endothelial cell dies and the blood-brain barrier is compromised, oligodendroglia can patch the damaged region.
Blood circulation
Astrocytes are activated by glutamate released in synapses, leading to the increase of intracellular calcium. The rise of calcium increases the levels of arachidonic acid, which is metabolized into prostaglandins and EETs (epoxyeicosatrienoic acid). Prostaglandins and EETs are vasoactive metabolites that produce vasodilation. There is another molecular pathway with opposite results: vasoconstriction. The arachidonic acid may be transformed into 20-HETE, which is a vasoconstrictor molecule. It may be that vasoconstriction or vasodilation depends on the nitric oxide released by neurons. Nitric oxide is a gas that influences the smooth muscle of arteries regulating the blood circulation.
Astrocytes are connected by gap junctions and form wide cellular networks. The cytoplasm of these astrocytes are connected forming a kind of syncytium, and large populations of astrocytes may be physiologically synchronized. The influence of these connected astrocytes on blood circulation may affect broad nervous areas
4. Pathology
Astrocytes are able to endure to low levels of oxygen and glucose. Under these conditions, they can stay alive longer than neurons because they are able to store glycogen and produce ATP from anaerobic metabolic pathways. Thus, astrocytes are important during extreme situations and certain pathologies. After brain damages, astrocytes become reactive (reactive astrocytes), that leads to hypertrophia or hyperplasia. This is called reactive gliosis. Two types of astrocytes can be distinguished during reactive gliosis. A2 astrocytes are mostly involved in repairing, whereas A1 astrocytes favor tissue degradation. A2 astrocytes are also important for angiogenesis (formation of blood vessels). Reactive gliosis is observed in Parkinson, Alzheimer and Huntington's chorea diseases. Furthermore, because they can divide, astrocytes are responsible for most brain tumors, known as glioma.
5. Others
Astrocytes provide neurotrophic molecules to neurons, are a major source of extracellular matrix, can act as detoxification centers by storing some metals and ammonia.
-
Bibliography ↷
-
Liddelow S, Barres B. 2015. SnapShot: Astrocytes in Health and Disease. Cell. 162: 1170-1170.e1.
Ransom B, Behar T, Nedergaard M. 2003. New roles for astrocytes (stars at last). Trends in neurosciences. 26: 520-522.
Tabata, H. 2015. Diverse subtypes of astrocytes and their development during corticogenesis. Frontiers in neurosciences. 9, 114.
-