Ihe extracellular matrix is an aqueous environment containing many proteins and carbohydrates. In animal tissues, collagen fibers, elastic fibers, glycoproteins, and a large amount of other molecules present in the extracellular matrix are among large non-branched polymers of carbohydrate. The carbohydrate polymers are glycosaminoglycans, also known as mucopolysaccharides. In plant tissues, the main extracellular carbohydrate is cellulose, a glucose polymer, but glycosaminoglycans are not present.
1. Glycosaminoglycans
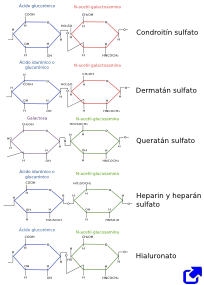
Glycosaminoglycans are unbranched carbohydrate polymers that can be very long molecules. They are composed of disaccharide repeating units containing an amine group on the first one (N-acetylgalactosamine or N-acetylglucosamine), whereas the second is usually galactose or glucuronic acid. Most of the hydration of extracellular matrix depends on glycosaminoglycans because water is strongly associated with the negative electric charges of their carboxyl groups (COO-) and sulfate groups (SO3-), (Figure 1). Glycosaminoglycan molecules occupy large volumes because their molecular secondary structure is quite inflexible providing, together with the high hydration, gel-like properties. Thus, it allows tissues with high proportion of glycosaminoglycans to resist mechanical loads. Furthermore, the diffusion of molecules through the extracellular matrix is highly facilitated. The most common glycosaminoglycans are hyaluronan and sulfated glycosaminoglycans, such as chondroitin sulfate, dermatan sulfate, keratan sulfate and heparan sulfate.
Hyaluronic acid
acid
Hyaluronan, also known as hyaluronic acid, is a distinct type of glycosaminoglycan. First, it does not bind chemically to other extracellular matrix molecules. Second, it is synthesized in the cell membrane, not in the Golgi apparatus. Third, it does not contain sulfate groups. Hyaluronan is made up of disaccharide units, D-glucuronic acid and N-acetyl-glucosamine, repeated up to 20000 times. It is usually associated to collagen and proteoglycans, and provides elasticity, lubrication and mechanical resistance. These properties are needed during development and in those places of the body where a high rate of cell proliferation occurs because hyaluronan makes easier the cell movement and molecule diffusion. As a large and rather inflexible molecule, hyaluronan occupies a large volume and provides many free spaces for diffusion. It is also abundant in those places with high friction forces such as joint cartilages.
Other glycosaminoglycans
All glycosaminoglycans, excepting hyaluronan, contain sulfate groups and are chemically bound to polypeptides. Proteoglycans are molecules consisting of a polypeptide chemically bound to one or more glycosaminoglycan molecules (see below). Chondroitin sulfate is a glycosaminoglycan composed of repetitions of N-acetylgalactosamine - glucuronic acid, and it is abundant in cartilage and nervous tissue Dermatan sulfate is made up of repetitions of glucuronic acid (or iduronic acid) plus N-acetyl-galactosamine. Keratan sulfate contains repetitions of N-acetyl-glucosamine and galactose, with different kind of sulfation depending on the monosaccharide. Heparan sulfate is synthesized by most cells, and is essential for the basal lamina. All these glycosamioglycans have many negative electrical charges that make frequent associations with chemocines or growth factors, laminins and fibronectins. Heparin is a heparan sulfate derivate release by mastocytes and basophil, used as anticoagulant. Heparin consists of repetitions of N-acetyl-glucosamine plus glucuronic acid (or iduronic acid), with different chemical bindings than those of hyaluronan.
2. Proteoglycans
Proteoglycans are molecules composed of a polypeptide and one or several sulfated glycosaminoglycans linked by chemical bindings (Figure 2). The chemical bond is through the serine amino acid. They are found in every animal tissue. Almost every cell may synthesize proteoglycans, and then they are released, inserted in the plasma membrane or stored in internals vesicles. Every cell is able to release different types of proteoglycans. Proteoglycans are synthesized and assembled within the cell. The polypeptide is synthesized in the endoplasmic reticulum, where some monosaccharides are also added. However, the elongation of the glycosaminoglycan chains, as well as the addition of sulfate groups, occurs in the trans domain of the Golgi complex. Most proteoglycans are exocytosed to the extracellular space, but some will be part of the plasma membrane, where they are inserted among the fatty acid chains of lipids thanks to a sequence of hydrophobic amino acids of the polypeptide
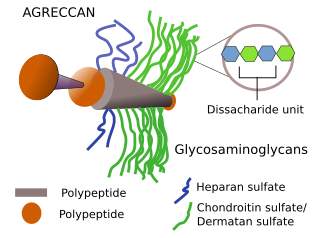
Different proteoglycans show different amino acid sequence and polypeptide length (ranging from 100 to 4000 amino acids). Only a few of polypetides may link glycosaminoglycans: about 17 different polypeptides link heparan sulfate, 20 link chondroitin/dermatan sulfate, and 8 link keratann sulfate. With these relatively low number of polypeptides, a wide variety of proteoglycans can be synthesized, since they can be bond to a different number and types of glycosaminoglycans. For example, decorin proteoglycan is made up of just one molecule of glycosaminoglycan bond to the polypeptide, whereas aggrecan proteoglycan contain more than 200 glycosaminoglycans. The number and distribution of sulfate groups in the glycosaminoglycan molecules also increase the diversity. For example, the sulfation depends on aging, tissue and pathology.
Proteoglycans can be classified in several families. Lecticans show a globular structure in the N-terminal part of the polypeptide, which interacts with hyaluronan a central domain with chondroitin sulfate, and C-terminal domain with type C lectin. They mainly contains chondroitin sulfate, and occasionally keratan sulfate. Aggrecan, versican, neurocan and brevican are members of lectican group. Aggrecan is abundant in cartilages, brevican and neurocan in the nervous tissue, and versican yin the connective tissue. The SLRP group of proteoglycans contains many leucine amino acids in the polypeptide, which is bound to chondroitin sulfate and keratan sulfate. Biblicane, decorin, fibromodulin and keratocan belong to this group. A function of SRLP proteoglycans is to estabilize collagen fibers. Another major group of proteoglycans contains heparan sulfate. Most of them are free in the extracellular matrix, such as perlecan and agrine. A few are associated to the cell membrane by the CD44v3 receptor, such as syndecan, and others, such as glypican, are inserted in the plasma membrane. A diverse group of proteins, such as CD44 receptor, amyloid precursor protein, and some collagen (IX, XII, XIV, XVIII), may or may not contain sulfated glycosaminoglycans, i.e., they are "part-time" glycosaminoglycans.
The biological activity of proteoglycans strongly depends on the molecular weight, number and disaccharide repeats, and the type of binding between saccharides of glycosaminoglycans. All glycosaminoglycans are negative charged, but the amount of negative charges is a major feature to understand their functions. The number of sulfate groups determines the overall negative charge, but how sulfate groups are sorted along the saccharide chain also determines the properties of the molecule and can make many different molecules. These functions are hydration, resistance to tensile forces, lubrication, help in cell physiology and motility, etcetera. For example, the mechanical role is essential in cartilage and joints. Furthermore, they act as anchoring points for cell attachment, either because the proteoglycan is part of the plasma membrane, or because they are recognized by cell adhesion molecules like integrins. It is curious that proteglycans are scarce in the stem cell niches.
Sometimes, proteoglycans can form chemical and physical barriers. Perineuronal nets are the regions of the extracellular matrix surrounding somata and proximal dendrites of neurons in the central nervous system. These extracellular coats may influence synaptic plasticity and formation of new synapses. Some neuropathologies are linked to damages in these nets. It is thought that perineuronal nets inhibit neuronal plasticity.
Sulfated glycosaminoglycans are places where some charged proteins may be transiently retained. Sometimes, the interaction of proteins with proteoglycans produces a conformational change that ends up in the activation or inactivation of the protein function. For example, heparin modifies antithrombin so that it can bind thrombin and X factor, resulting in inhibition of blood coagulation. Another example is the conformational change of fibronectin, that after the interaction with glycosaminoglycans, exposes molecular domains that interact with other extracellular matrix molecules. Another function of glycosominoglycans, as co-receptors, is to increase the affinity between ligand and receptor.
3. Cellulose
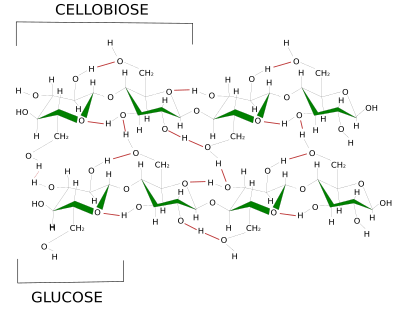
Cellulose is the main component of cell walls, the extracellular matrix of plants. Some authors do not include cell wall as a type of extracellular matrix, but here it will be studied as a highly specialized extracellular matrix. Cellulose forms a paracrystalline structure consisting of polysaccharides made up of glucose molecules (more than 500 per cellulose molecule) linked by β(1-4) bonds (Figure 3 and this figure). About 50 cellulose molecules join together by hydrogen bonds and van der Waals forces, forming structures known as microfibrils, which are oriented with the same polarity. Cellulose microfibrils are laterally linked together by hemicellulose and pectins to form cellulose fibrils and fibers. Fibers can be visualized at light microscope.
In a similar way as hyaluronan, cellulose is synthesized in the plasma membrane by the cellulose synthase enzyme, a transmembrane protein with 8 intramembrane segments (Figure 4). It takes glucose units from cytosol, makes them cross the plasma membrane, and chemically links them in the extracellular space. Up to 36 cellulose synthase molecules get together to form a macromolecular complex known as cellulose synthase complex, which can synthesize up to 36 cellulose molecules at the same time. As they are synthesized, cellulose molecules associate into cellulose microfibrils.
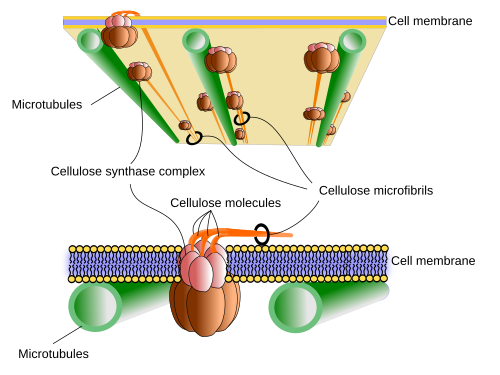
An interesting question is how cellulose strands are laid over the cell membrane because the orientation of the microfibrils determines the orientation of cellulose fibrils. The growth of the body and the organs of plants is mostly a consequence of the increase of cells size, which is produced by hydrostatic pressure. Plant cells grow in the direction with a lower mechanical strength of the cell wall. The cell wall resistance is lower along the axis perpendicular to the cellulose fibrils. The orientation of cellulose microfibrils over the cell membrane is determined by the movement of the cellulose synthase during the synthesis of the cellulose molecules, which in turn depends on the orientation of the underlying microtubules located just under the cell membrane (Figure 4). Microtubules are like fences that cannot be crossed by the large cellulose synthase complexes, as these enzymes move while synthesizing cellulose molecules. Thus, by restraining the movements to certain directions, microtubules are responsible for the spatial orientation of cellulose fibers. There are other extracellular and intracellular factors influencing the movement of the cellulose synthase complexes. In this way, plants can control the growing direction of stems, leaves, and other organs of the body depending on the light, gravity or water. Besides the orientation of the cellulose fibers, cells can regulate the strength of the cell wall by other mechanisms. They can modify molecules like pectins or increase the acidification, that result in lower mechanical resistance places, so that cells enlarge through those areas.
-
Bibliography ↷
-
Bibliography
Bosman FT, Stamenkovic I. 2003. Functional structure and composition of the extracellular matrix. Journal of pathology. 200: 423-428.
Lamoureux F. 2007. Proteoglycans: key partners in bone cell biology. BioEssays. 29: 758-771.
McFarlane HE, Döring A, Persson S. 2014. The cell biology of the cellulose synthesis. Annual review of plant biology. 65: 69-94.
Soares da Costa D, Reis RL, Pashkuleva I. 2017. Sulfation of Glycosaminoglycans and Its Implications in Human Health and Disorders. Annual review of biomedical engineering. 19: 1-26.
-
 Structural proteins
Structural proteins 