1. Cell wall
- Layers
- Components
2. Basal lamina
3. Connective
4. Tendon
5. Cartilage
6. Bone
7. Nervous
8. Blood
Many tissular functions rely on the extracellular matrix properties, which in turn depend on the content of molecular types and proportions. This is true for both plant and animal tissues. The extracellular matrix of plants is known as cell wall. Here, we are going to include the cell wall as a type of extracellular matrix, although animal and plant cell extracellular matrices are really different.
1. Cell wall
Plant cells can not be understood without the cell wall, which is a distinct feature when compared with animal cells. Cell wall provides structural support and protection to plant cells, and therefore to the entire plant body. It also determines the shape and size of the cells and organs, and it is responsible for tissue features like resistance to mechanical forces, both loads and stretching. The aerial parts of plants are possible because of the plant cell. The supporting role of cell wall in the plant body continues even after cells die. For example, wood is mostly cell walls. The most prominent component of cell wall is cellulose, which is the most abundant molecule in nature. Depending on the plant tissue and differentiation state of the cell, cell wall may show different molecular composition.
Layers
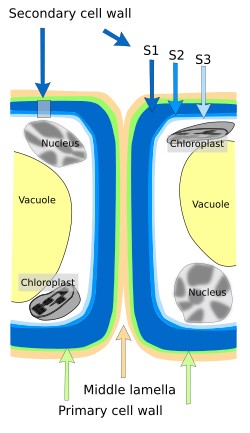
The thickness of the cell wall depends on the type and age of the cell. Up to 3 layers can be found: middle lamella, primary cell wall, and secondary cell wall (Figure 1). Cells synthesize these layers starting with middle lamella, then primary cell wall, and finally secondary cell wall, which is not always present. The most recent layer is the nearest to the plasma membrane. All plant cells have a middle lamella, which is shared with the neighboring cells, and a primary cell wall with variable thickness, but only some cells develop secondary cell wall. Primary cell wall allows cell to grow in size because it can be stretched by hydrostatic pressure, which is referred as turgency. Secondary cell wall is synthesized by cells that withstand heavy mechanical loads and by some vascular cells. Secondary cell wall may be divided in sublayers (S1, S2 and S3). The synthesis of secondary cell wall usually means that cell is not going to grow anymore, because cell elongation is not possible. Secondary wall is no permeable to water but there are many pores (pits) that allow water to cross the secondary cell walls of adjoining cells.
Components

Three types of polysaccharides are the main components of the cell wall: cellulose, hemicellulose and pectin (Figure 2). There are also proteins and ions. Cellulose is the main component of the cell wall. It is a polysaccharide made up of D-glucose units linked by β (1-4) bonds. Long chains of cellulose are synthesized in the plasma membrane and parallel-oriented to form cellulose microfibrils with variable diameters, which in turn associate to form cellulose fibers. These fibers are visible at light microscopy and are very resistant to mechanical stress, similar to steel. Cellulose fibers also contain other saccharides like hemicellulose and pectin, as well as glycoproteins. Hemicellulose is a family of polysaccharides containing xylose, glucose and mannose, which make cross-bridge bonds with cellulose microfibrils, strengthening the network of cellulose fibers. Pectins are the more diverse type of polysaccharides that can be found in primary cell wall, but they are absent from secondary cell wall. They are heterogeneous polysaccharides, contain galacturonic acid, and make cross-bridge links between cellulose microfibrils. Pectins are highly hydrophilic molecules.
Other molecules can be found in the cell wall of particular cell types. Callose is a polysaccharide located between the cell membrane and the cellulose part of the cell wall. It is synthesized in the plasma membrane. Callose is a lineal polymer of glucose linked by β(1-3) bonds that does not form semi-crystal structures. Lignin is a complex polyphenolic polymer released into the cell wall after the secondary cell wall is synthesized. It restricts the water diffusion and provides a great mechanical strength. The cell wall of some cells, like epidermal cells, shows cutin and suberin, which are lipids preventing water lost and the entrance of pathogens. Enzymes can also be found in the cell wall, contributing to cell wall remodeling. Glycoproteins are involved int the molecular network that provides structural stability of the cell wall.
2. Basal lamina
The basal lamina is a thin layer of extracellular matrix found in the basal part of epithelia, and around muscle cells, adipocytes and nerve cells of the peripheral nervous system. It works as a physical support and as a selective permeable barrier. The basal lamina appears early during the embryo development to separate and support the first tissues. In adult kidney glomeruli, it is a key player in filtering the blood. The basal lamina and basal membrane are sometimes confused in some tissues, such as muscle tissue. Basal membrane is actually the basal lamina plus a more external fibrillar and reticular layer. The basal lamina is composed of molecules arranged in an ordered 3D network. It contains type IV and VII collagen, laminin, perlecan proteoglycan, and nidogen protein. Basal lamina is linked to the membrane of the basal domain of epithelial cells by connections between integrins and laminins.
3. Connective proper
The extracellular matrix of loose connective proper tissue shows low protein density, and a higher proportion of hyaluronan and proteoglycans than collagen and elastic fibers. The main role of this extracellular matrix is to fill and hydrate intercellular spaces and provide the environment through which many cells travel from one site to another. The fibroblast is the typical cell type of connective proper tissues. In the dense connective tissue there are many collagen fibers, either arranged parallel to the tensile forces, such as in tendons, or disorganized as in the dermis and in the intestine. Elastic fibers may be abundant, as in the artery wall.
4. Tendon
Tendon clearly shows that strength and elasticity of tissues depend on the features of the extracellular matrix. Collagen fibers of tendons are oriented parallel to the direction of the mechanical stress, which is only in one axis. The tendon extracellular matrix is rich in collagen fibers made up of collagen types I, II and III, which are up to 65-80 % of the dry weight, whereas elastin is only 1-2 %. Collagen and elastic fibers are embedded in a rich mieliu of proteglicans. Fibroblasts, also referred to as tenoblasts, are found among the collagen and elastic fibers.
In tendons, collagen molecules are organized in three levels. The first level consists of microfibrils (4 nm in diameter) around the cells. These microfibrils are semi-flexible, and they can be reoriented according to the tensile direction. In the second level, microfibrils join form fibrils. The third level is the formation of fibers by growing and association of fibrils. Fibers may be 1 to 20 µm in diameter and associate to form sub-fascicles, also containing fibroblasts. Around sub-fascicles, there are connective tissue with nerve cells, blood and lymphatic vessels. Sub-fascicles may be 15 to 500 µm in diameter, and join to form fascicles of 150 to 1000 µm in diameter. Fascicles associate to form tertiary fascicles of 1000 to 3000 µm. Along the tendon, glycoproteins, glycosaminoglycans and other molecules associate with fibrils and fibers to stabilize the whole structure.
5. Cartilage
Cartilage extracellular matrix is synthesized by chondrocytes, and is responsible for both stiffness and elasticity of cartilage. It is mainly composed of type II collagen, up to 25 % of dry weight. Collagen types IX and XI are also present in lower proportion. The second most abundant molecules are glycosaminoglycans, such as hyaluronan, and proteolycans such as aggrecan, which join to form macromolecular complexes. Collagen counteracts tensile loads and glycosaminoglycans dampen mechanical pressures. Elastic cartilage contains abundant elastic fibers, which are necessary for the elasticity of structures like pharynx, epiglottis, and pinna.
6. Bone
The bone extracellular matrix contains type I collagen intermingled with a network of calcium phosphate crystals (which is up to 70 % of the bone dry weight). Collagen allows bone to be elastic enough to avoid bone fragility and not to be easily broken, and calcium phosphate crystal provides stiffness and hardness. There are also proteoglycans and glycoproteins, which are less abundant, but very important for the organization of collagen fibers, mineralization and resorption of bone. Chondroitin sulfate accounts for 67-97 % of the bone glycosaminoglycans.
7. Nervous tissue
There is little amount of extracellular matrix in the nervous tissue. Hyaluronic acid and proteoglycans are more abundant than collagen, elastin and glycoproteins. During development, extracellular matrix is synthesized and released by neurons and glial cells. The role of hyaluronic acid is structural and, together with other molecules, helps to form molecular scaffolds made up of hyaluronic acid, tenascin and proteoglycans. There are up to 16 types of chondroitin sulfate in these molecular networks. The composition of perineural nets changes during development. During the first weeks of development of mammalian embryo, they contain neurocan, versican and tenascine C, whereas in adults there is more content of brevican and aggrecan. In addition, type C chondroitin sulfate is abundant in embryonary nervous tissue, but gradually decreases after birth and disappears in adults, where it is replaced by type A chondroitin sulfate. During development, proteoglycans that form these networks are larger and form less compact extracellular matrix, so that neuronal sprouting and plasticity is favored. However, in adults, smaller proteoglycans form more compact extracellular matrix that makes more difficult neuronal plasticity.
Around neurons, dendrites and axonal initial segments, there is a thin limiting layer of extracellular matrix referred to as perineuronal net. It shows a more regular organization than the rest of the extracellular matrix. Perineuronal nets consists of hyaluronic acid, proteoglycans with chondroitin sulfate and tenascin R, organized as trimeric structures linked to one another. The appearance of the perineuronal nets is at same time as the decline of nervous tissue plasticity, at the end of its development. In turn, neuronal plasticity is reactivated when perineuronal nets are removed or weakened. They also seem to inhibit cell movement and remodeling.
8. Blood serum
Blood serum as a type of extracellular matrix is not accepted by everyone, but, here, it is considered as a highly specialized extracellular matrix, containing up to 90 % of water. It is the environment of blood cells. The most abundant protein of blood serum is albumin, which maintains a physiological osmotic pressure between blood vessels and tissues, preventing edemas. Other proteins are γ-immunoglobulins, which are the immune system antibodies, α- and β-globulins, which are carriers of elements such as copper and iron. Fibronectins can move between tissues and blood serum. Fibrinogen is essential for blood coagulation. Other blood serum molecules show low molecular weight, and they can be found in the extracellular matrix of other tissues since they can freely cross endothelium.
Bibliografía
McFarlane HE, Döring A, Persson S. 2014. The cell biology of the cellulose synthesis. Annual review of plant biology. 65:69-94.
 Glycoproteins
Glycoproteins 