1. Living organism
2. Where?
3. When?
4. How?
- Organic molecules
- Polymers
- Cell membrane
- Autoreplication
- Molecular interactions
- Genetic code
- DNA
The discovery of the origin of life is the discovery of the origin of the cell. It is unknown how the first cell appeared on Earth, but it is widely accepted that it was by physicochemical processes. In the twenties of the 20th century, A.I. Oparin and J.B.S. Haldane proposed the first physicochemical theories on the origin of life (also suggested by C. Darwin in one of his personal letters). The development of theories about the emergence of the first cells is based on hypothesis and in laboratory experiments that simulate the conditions thought to be present during the origin of life on Earth.
The origin of life as a physicochemical process has consequences in the field of biology. a) We can create life. A cell could be made from scratch by using molecules that exist in current cells and placing them together into a membrane bound compartment. A branch of biology, known as synthetic biology, is currently undertaking the first serious attempts to make a new cell from just molecules. The whole DNA of a prokaryotic cell has already been synthesized in the laboratory, introduced into a cytoplasm, and a new functional new cell has been obtained. A complete eukaryotic chromosome has also been synthesized. b) Extra-terrestrial life. Physicochemical conditions, similar to those present on Earth during the origin of life, may have occurred elsewhere in the Universe. Thus, life forms could have appeared in the past and may exist today in other parts of the Universe. Extraterrestrial life may have emerged in many planets and many times, and perhaps it is happening right now.
1. What is a living organism?
When searching for the origin of life, it is first necessary to know what a living organism is. We are all able to recognize a living organism, but it is more complicate to write a definition of what a living being is. It could be said that life is one of the properties that a living organism must have. However, there is a new problem: what is life? There is no one definition of life that is widely accepted by the scientific community. It is a paradox that Biology, part of the science dealing with living organisms, is studying something that is not yet precisely defined. The perception of life is just a feeling. Nowadays, scientists often define life not with a single statement, but as a set of properties that an organism should fulfill to be considered as a living organism. Again, there is no general agreement on how many and what these properties should be, but some of the most popular are the following:
a) Reproduction and transmission of information encoded by deoxyribonucleic acid (DNA).
b) Maintaining homeostasis by using external energy (metabolism).
c) Ability to respond to external and internal stimuli.
d) Ability to evolve by Darwinian evolution (variation and natural selection).
e) Some others.
The lack of a precise definition of life undermines the search for life in other planets. We intuitively know what we are searching for: something similar to what we have seen on Earth, but not something that fits in a definition of life or of a living being. So, if there are organisms in very unusual forms in other planets, we may overlook them or we may not be able to classify them as living beings.
2. Where did the first cells appear?
Scientists agree that the first cells emerged from organic molecules in the early Earth. These molecules may have been generated in different terrestrial environments and accumulated in the water. However, it is not excluded that many of these early organic molecules were synthesized in other planets and in the outer space, and came to the Earth as rains of asteroids and comets. Organic molecules were concentrated in some places where it is supposed that the first cells emerged. It has been suggested that these place were near to hydrothermal vents in the ocean or hot springs in the sweet water, with a rich mineral content. Hydrothermal vents show temperature and ions gradients that favor mineral catalysis to form complex molecular systems that evolved into the first cells.
3. When did the first cells appear?
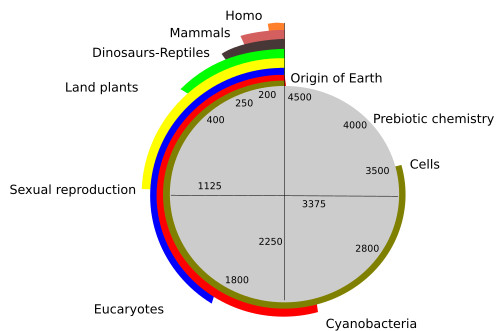
The Earth is about 4500 x 106 years old. The fossils indicate that the first cells were already on Earth between 3500 x 106 and 3800 x 106 years ago (Figure 1). During the first 500 x 106 years, the environmental conditions were not suitable for living forms due to high temperature, lack of protective atmosphere, and intense “rain” of meteorites. However, from 1000 to 1200 x 106 years later, the first cells could have left sedimentary deposits and organic molecules as a consequence of their metabolism. It means that the physicochemical processes leading to the first living organisms should have started earlier, in a period called the prebiotic era.
4. How did the first cell appear?
We can imagine the steps, starting from simple molecules until the appearance of the first cells. There is no consensus on how many steps there were, what the sequence was, or how these steps occurred, but the main stages during the origin of cells may have been as follows:
Organic molecules
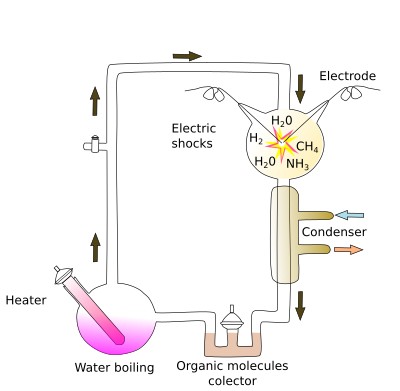
Organic molecules, plus water and some ions, are the building blocks of living organisms. The most relevant are proteins, nucleic acids, sugars and fat. How were they synthesized in the early Earth? There are several plausible scenarios. a) Terrestrial origin under extreme environmental conditions. If a flask containing CO2, ammonia, methane and hydrogen, is heated to high temperature and exposed to electric discharges, some complex organic molecules are obtained, such as hydrogen cyanide, formaldehyde, some amino acids, some sugars, purines and pyrimidines (necessary for nucleotide synthesis). This was the experiment carried out by Miller and Urey when they were trying to replicate the earliest environment on Earth (Figure 2). It does not prove that the origin of life was like this. However, it shows that complex organic molecules can be formed by physicochemical reactions. Furthermore, different places on early Earth could have had distinct environmental features that yielded different sets of complex organic molecules. Plausible sites for prebiotic synthesis are hot sea vents, fumaroles and hot springs, where strong temperature gradients and high water pressure can be found, in addition to minerals. b) Extraterrestrial origin. It has been shown that more or less complex organic molecules can be synthesized in the outer space, and they can be found in comets and meteorites. It is possible that a huge amount of extraterrestrial organic compounds reached the Earth surface, which could have then started the chemical reactions for the origin of life.
Polymers
So, we already have organic molecules. However, the most relevant molecules for the cell are present as organic polymers: amino acid chains for proteins, nucleotides strands for DNA and RNA, sugar chains for starch and glycogen. In the early Earth, to get these polymers was a problem because joining molecular units to make long chains is not an easy task. A good polymerization system that could have been worked properly at the origin of life has yet to be found. However, several hypotheses have been proposed.
a) Heating and drying. In the laboratory, heating semi-dry compounds have been shown to produce chains of organic molecules.
b) Mineral surface catalysis. Minerals could have been key players during the origin of life for several reasons. They concentrate, select, work as template, and perform catalysis of organic chemical reactions. They may have work in the outer space, other planets and on the Earth surface. The early Earth had a wide variety of mineral types to catalyze and yield different complex organic compounds. More than 420 mineral types are thought to be present in the early Earth. Catalysis by mineral surfaces, such as polyphosphates and clays, and other catalytic minerals, produces polymers having randomly ordered units. It has been proved that clays are able to recruit organic molecules and yield polymers, such as RNA strands. In an adverse environment, minerals may also have been sheltered places for the first complex organic molecule systems.
c) Fumaroles and hot springs. Sea vents, fumaroles, hot springs, hydrothermal vents, provide strong gradients of temperature and water pressure that, with the help of minerals, facilitate chemical reactions. Currently, organic molecules are produced in fumaroles. Although fumaroles are likely places to produce organic compounds, fresh water hydrothermal vents are also places of interest, since they are near volcanoes where hydration and drying cycles can concentrate organic compounds and facilitate chemical reactions at high temperatures. Furthermore, freshwater environments are more suitable for spontaneous membrane assembly than the sea, and there are low concentrations of calcium and magnesium, ions that inhibit the formation and integrity of membranes.
e) Lipid membranes. Several laboratory experiments have demonstrated that the surface of lipid membranes, such as cell membranes, can recruit, select and concentrate simple organic molecules. In membranes, organic molecules are close to one another, and the lipid environment can facilitate chemical reactions, such as those involving nucleotides and amino acids. This scenario is interesting because it addresses two questions: why only a few specific types of organic molecules were included in membrane bags or vesicles, and how the protocells were first formed.
Cell membrane
One of the major leaps during the birth of the first cells was the development of a barrier to separate the intracellular and extracellular environments. Membranes provide many advantages: a) molecules for metabolic reactions are held together and are not lost by diffusion; therefore, the chance of chemical reactions is higher and more efficient; b) internal molecules are not shared with neighbors, so that new advantageous molecules for new chemical pathways are not used by competitors, that is, "selfish evolution"; c) a proper internal environment can be set to enhance chemical reactions and to counteract or buffer external environmental changes as well. Maintaining optimal inner parameters is known as homeostasis. Lipid membranes can spontaneously assemble from amphipathic lipids, which have hydrophilic and hydrophobic domains. However, the ancient membrane lipids could have been different from nowadays ones because the current membrane lipids require a complex metabolic pathway to be synthesized. Whatever they were, when added to aqueous solutions, and after applying mechanical forces to the solution, amphipathic lipids spontaneously formed thin layers similar to cell membranes. Current membrane lipids have two fatty acid chains. This feature allows the formation of membranes at a micromolar concentration of lipids. Single-chain fatty acid lipids require millimolar concentrations to spontaneously form a membrane. Furthermore, 10 to 14 carbons in fatty acid chains make the membrane structure more stable. It is possible to regulate membrane fluidity by changing the number of double bonds between the carbons and by changing the concentration of sterols. It is unknown what type of lipids were the components of the first membranes, but the current membranes are all made up of glycerophospholipids and sphingolipids, as well as sterols.
Two ways for the association between organic molecules, such as nucleotides and amino acids, and membranes have been suggested (Figure 3). a) It is plausible that water shaking could have led membranes sheets to form vesicles or small bags that enclosed groups of molecules. By chemical reactions, these molecules increased in number and the vesicle became larger. At one point, the vesicles gained the ability to divide by strangulation, giving rise to two new vesicles with the same types of molecules as the mother vesicle. The growth of the vesicles could have happened thanks to the properties of the membrane: allowing the extracellular supply of small molecules that could freely cross the membrane, but hampering the exit of larger molecules (such as polymers). b) Another scenario suggests that there was an association between simple organic molecules and lipid membrane surfaces. It has been shown that membranes recruit and increase the concentration of organic molecules on their surfaces and facilitate chemical reactions. The chemical reactions in 2D environments are more efficient than in 3D ones. Thus, polymers (oligopeptides and oligonucleotides) originated in close association with membrane surfaces and gained complexity over time. At one point, these macromolecules became trapped into vesicles or, if synthesized onto vesicles membranes, they acquired the ability to cross the membrane and remain within the vesicle. The vesicle growing and strangling may have happened later in a controlled manner. If one of these mechanism actually occurred, it is convenient to reconsider the order of events during the cell birth, because membranes were then the most important elements during the formation of the first protocells.

Autoreplication
Another major step during the origin of life was to change from a system of molecules formed randomly by external agents to a system with molecules generated by the system itself. It was then possible to produce copies of the molecules of the system, and therefore to achieve self-replication. It led to a main property of life: the transmission of information. There are two types of information that can be transmitted by the self-replication of molecular polymers: the particular sequence of monomers and the spatial organization of the newly synthesized molecule (the 3D structure depends on the sequence of monomers). Do we have already a genotype and a phenotype? The monomers and energy to synthetize these polymers were taken from the environment because they could cross membranes. Within vesicles, more or less accurate copies of some polymer systems were made. However, the self-replication process could make mistakes leading to variations of the monomer sequences. Some polymers or polymers system with slightly different sequences could have performed better during their own replication and yield more copies. Then, membrane vesicles with more productive molecules could grow faster, get more external resources and leave more offspring. Thus, we have a competition for external resources by populations of vesicles having different sets of polymers. They had discovered another property of life: variability and natural selection, i.e.,Darwinian evolution.
Some authors think that one type of molecule could have initiated the autoreplication and evolution process, but others suggest that a system of molecules connected by chemical reactions could have been the starting point, with a self-replication of the entire molecular network. Thus, two main model have emerged to explain the prebiotic process that led to the first cell: the ARN world and the metabolic world.
ARN World. If a single type of molecule is assumed to be the first self-replicator, which one was it? DNA is not a very reactive molecule and must be “manipulated” by proteins, which are the true workers of cells. In the current cells, proteins are synthesized from DNA-based information and DNA needs proteins. Then, which came first, proteins or DNA? The eyes turn to the RNA. Some RNA molecules have the ability to carry out enzymatic reactions (that is why they are known as ribozymes). For example, ribonucleoproteins can cut messenger RNA molecules and join them (mRNA splicing), and protein synthesis on ribosomes is accomplished by ribosomal RNA. Therefore, it is plausible, although unlikely, that RNA molecules could have made copies of themselves in the early Earth, by synthesizing a sequence complementary to their own nucleotide sequence. The nucleotide sequence is known to determine the 3D form of RNA, influencing stability and activity (Figure 4). Thus, sequence and spatial conformations may have worked together to make molecules more competitive by increasing their stability and yielding more copies. In this scenario, some errors may have occurred during the copying process ("mutations") that gave more or less stable molecules. Darwinian competition could have then started. Over time, the vesicles were enriched with those RNAs that made copies more efficiently. Thus, sequence (genotype?) and spatial conformation (phenotype?) could provide profitable features. All these steps have been proposed to occur during the prebiotic period on early Earth, and are the base of the RNA world model.

Metabolic world. An early world with networks of chemical reactions that gave rise to complex molecular systems also has some experimental support. In this model, replication was not achieved by a single type of molecule, but by a group of molecules connected by chemical reactions. At some point, a membrane was also needed for creating a protected environment, got the ability of growing and division of the system, and the ability to evolve. Authors supporting the metabolic world do not discard a major role for RNA during the origin of life. However, RNA could have participated during later steps in the evolution of these molecular systems. In fact, some authors suggest that RNA would initially be a parasite of these chemical systems. Eventually, the RNA took control of the system.
Molecular interactions
No matter the molecule, or molecules, that got the capacity of self-replication and evolution, interactions between different types of molecules (polypeptides, DNA, RNA, lipids, sugars) should have occurred, leading to complex chemical systems. It can be imagined that the association of two types of molecules, let's say RNA and some polypeptides, would have ended up being beneficial for both. Later, the complexity increased and new molecules, such as DNA, became integrated into the net of chemical interactions. Over the time, the entire molecular mix evolved and faced environmental selection by modifying interactions between each other. This introduces a new concept: coevolution of molecular forms. This could have happened 3500-4000 x 106 years ago.
Genetic code
At some point in this story, RNA must have been involved in the synthesis of proteins (polypeptides). This was a critical step because a code was invented: 3 nucleotides in a particular order meaning a particular amino acid. This code is known as the genetic code, and it is present in all living organisms. Every cell studied so far shows the same nucleotide triplets for the same amino acids (with some minor exceptions). The universality of the genetic code suggests that it was invented only once, and was inherited by all living organisms. This reasoning leads to think that all living organisms descend from only one type of cell, known as LUCA (last universal common ancestor). However, some authors suggest that the genetic code was invented before the cellularization process, and although bacteria and archaea inherited the genetic code, they went to independent cellularization processes.
DNA
In current cells, the information inherited is encoded in DNA, not in RNA, neither in proteins. Compared to RNA, DNA is a more stable molecule because it is a double strand, which makes easier replication and repairing. There is a type of enzymes, known as reverse transcriptases, with the ability to transform RNA sequences into DNA sequences. Many viruses, such as the AIDS virus, contain this enzyme to convert information of RNA (present in the infectious phase: virion) into DNA, which can be replicated within the host cell. At some point before LUCA, genetic information was trasferred from RNA to DNA, which became the container for storing, transcribing and transmitting the information necessary to form a new protocell.
There are many uncertainties and controversies about each one of the above steps, and others not mentioned here. How did the events happen?, what were the most important molecules?, what was the real environment in every step?, and many more questions are still under a heated debate. There is no doubt that knowing how was the origin of life, i.e., the origin of the first cells, is one of the main scientific challenges.
-
Bibliografía ↷
-
Bibliografía
Black RA, Blosser MC. 2016. A self-assembled aggregate composed of a fatty acid membrane and the building blocks of biological polymers provides a first step in the emergence of protocells. Life. 6: 33
.
Michalak R. 2006. RNA world - the dark matter of evolutionary genomics. Journal of evolution biology. 19(6): 1768-1774.
Müller UF. 2006. Recreating an RNA world. Cell and molecular life science. 63: 1278-1293.
Oparin AI. 1970. Origen de la vida en la Tierra. Editorial Tecnos S.A. Traducción de la tercera edición rusa.
Orgel LE. 1994. Origen de la vida sobre la Tierra. Investigación y Ciencia. 219: 47-53.
Peretó J. 2005. Controversies on the origin of life. International microbiology. 8: 23-31.
Robinson R. 2005. Jump-starting a cellular world: Investigating the origin of life, from soup to networks. PLoS Biology. 3(11): e396.
Shapiro R. 2007. El origen de la vida. Investigación y Ciencia. 371: 18-25.
Warmflash D, Weiss B. 2006. ¿Vino de otro mundo la vida?. Investigación y Ciencia. 352: 24-31.
-
 Cell theory
Cell theory