Immunohistochemistry is a technique for studying the tissular location of particular molecules by using antibodies as tools. There is a broad variety of commercial antibodies, and the standardization of the protocol has made immunohistochemistry a straightforward and powerful lab technique. It relies on the high affinity and specificity of the antibody-antigen recognition. To visualize the antibody-antigen binding, antibodies are conjugated with enzymes or fluorescent molecules that amplify enormously the detection threshold.
Antibodies used in immunohistochemistry are G type immunoglobulins, produced by the B lymphocytes of the immune system. There is a massive production of G immunoglobulins when an animal is inoculated with a molecule that is recognized as foreign by the immune system. These immunoglobulins are released and found in the blood. They are then are purified from blood serum and used for immunohistochemistry.
Complex molecules like proteins may have several antigenic determinant sites, that is, molecular domains capable of starting an immune response. An antigenic determinant activates a B lymphocyte clone, or lineage, that produces the same type of G immunoglobulin against that molecular domain. Immunoglobulins from all activated B lymphocyte lineages after the injection of a complex molecule are found in the blood serum. A polyclonal antibody is the purified serum containing the immunoglobulins from all the activated B lymphocyte clones (Figure 1). On the other hand, cells from a particular activated clone can be isolated from the animal body and cultured in vitro. Each colony of each single in vitro culture contains B lymphocytes that produce the same G immunoglobulin, which recognizes one antigen determinant of the injected molecule. The supernatant of each culture can be purified, which is therefore known as monoclonal antibody.
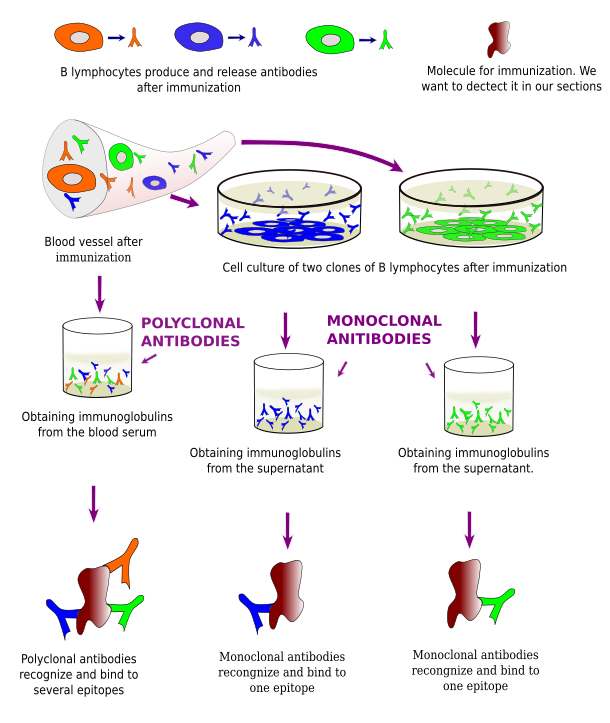
G immunoglobulins have two molecular domains or regions: variable and crystallizable. The variable region (Fab) is responsible for recognizing the antigenic determinant, or epitope, in the foreign molecule. Each G immunoglobulin molecule has two equal variable domains, i.e., they recognize the same antigenic determinant, so that each single G immunoglobulin may bind two foreign molecules at the same time. Crystallizable regions are similar for all G immunoglobulins produced by individuals of the same species.
If the epitope is modified, the antigen-antibody reaction will be weak or not happening at all because the antibody is not able to recognize the epitope. Thus, the fixative and fixation method have to be chosen carefully to preserve the features of the epitope in the tissue. Different fixatives are used depending on the nature of molecule to be detected. In addition, embedding may add damages to epitopes; for example, paraffin embedding is not recommended for the detection of some molecules because dehydration and heat may harm the antigen-antibody reaction. The best preservation for most epitopes is achieved in frozen and vibratome sections, after a proper fixation.
Immunoglobulins are molecules that cannot be visualized with a light microscope. Therefore, they must be conjugated (bound) to other molecules that make possible visualizing the antigen-antibody reaction (Figure 2). There are two types of molecules that are commonly conjugated with antibodies: fluorescent molecules and enzymes. Fluorescent molecules can be directly visualized with fluorescent microscopes, while the enzyme activity produces colored and insoluble products that can be observed at light microscopy. In both cases, the signal (what we can observe) are located where the antigen-antibody reaction occurred.
Fluorescent-conjugated immunoglobulins have advantages that we will see below. However, one disadvantage is that the fluorescence of the fluorescent-molecule is exhausted with time. On the other hand, sections processed for immunohistochemistry, i.e., using enzymes, can be dehydrated, mounted, and maintained nearly forever. Peroxidase and alkaline phosphatase are the typical enzymes that are conjugated with immunoglobulins.
Direct immunohistochemistry (Figure 2) means that the primary antibody is conjugated with a molecule, either an enzyme or a fluorescent molecule. Nowadays, indirect immunohistochemistry is more common (Figure 2). It means that the primary antibody is not conjugated with any molecule and there are other molecules between the primary antibody and the conjugated molecule.
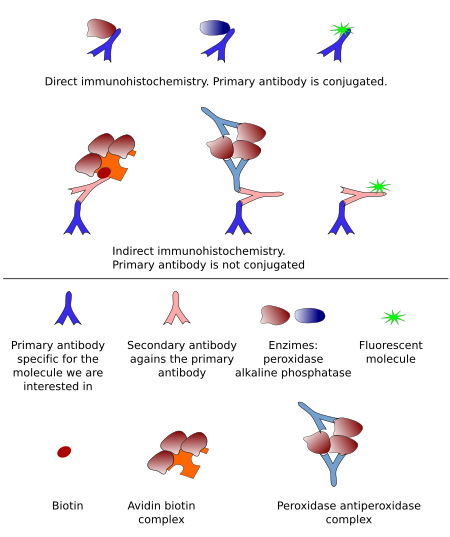
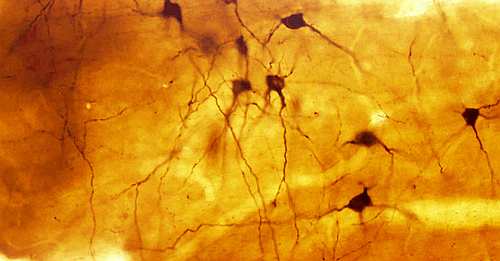
Immunodetection based on enzyme activity is known as immunohistochemistry, or immunocytochemistry. Direct immunohistochemistry was developed first, and later it came the peroxidase-anti-peroxidase indirect method (Figure 2). Nowadays, biotin-conjugated secondary antibody combined with avidin-biotin-peroxidase or streptavidin-peroxidase is the most common method (Figure 3). Indirect methods provide higher versatility and more sensitivity (more signal for an amount of epitopes). The general procedure begins with sections from fixed material . These sections are incubated in a blocking solution containing high protein concentration (usually bovine serum albumin) to prevent non-specific binding of immunoglobulins. Then, sections are incubated in the primary antibody, and subsequently in biotin-conjugated antibody, and in the enzyme complex. Finally, the enzyme activity is developed with its substrate and a substance, such as diaminobencidine for peroxidase or nitroblue tetrazolium for alkaline phosphatase, that is reduced and precipitates as a colored complex (Figure 4).

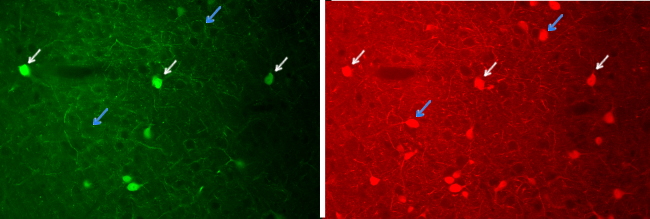
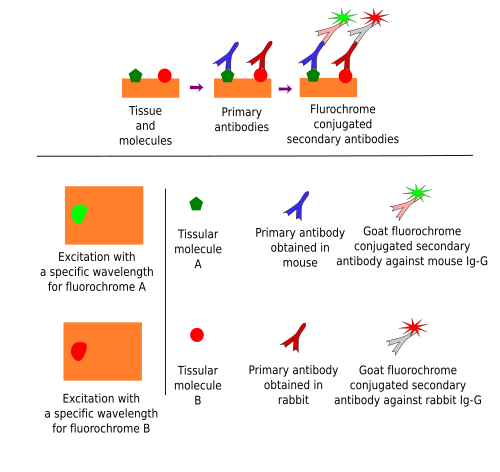
Immunofluorescence uses fluorochrome-conjugated immunoglobulins. To be precise, immunofluorescence is not immunohistochemistry because there is no chemical reaction. It takes advantage of fluorochromes, molecules that emit visible light when stimulated by a specific light wavelength. Nowadays, the indirect method is the most used (Figure 5). Although immunofluorescence can be used to study just one molecule, its real power is for detecting multiple molecules in the same cells or tissues, that is, co-localization of molecules (Figure 6). It is possible because different fluorochromes are excited in particular wavelength ranges so that two or more fluorochromes can be specifically excited by using a set of filters that let pass selected wavelength intervals. Pictures can be taken after excitation with each wavelength and the images are overlapped to study if an immunopositive structure, for example a cell, is labeled in more than one image, which means that there is co-localization (Figure 6).
 Lectins
Lectins