LLipid droplets were observed and described in the XIXth century, and they were named liposomes for many years. They were also known as lipid bodies, fat bodies, oil bodies, spherosomes and adiposomes. Most animal cells store lipids as droplets scattered in the cytoplasm. They can also be found in plant cells and even in yeast and bacteria. In fact, lipid droplets were first studied in plant seed cells.
Although the majority of cells may contain lipid droplets, adipocytes are animal cells specialized in producing and keeping lipid drops. There are two main types of adipocytes. Unilocular adipocytes (white fat cells) contain one very large lipid droplet that occupies most of the cytoplasm. The main function of white fat is to store and release energy when needed. Multilocular adipocytes (brown fat cells) contain multiple small lipid droplets in the cytoplasm and are specialized in producing heat. In animals, after the fat tissue, the second place where most fat is stored is the liver. There are many lipid droplets in the enterocytes of the intestine after the digestive process. In these cells, the triacylglycerols are exported as chylomicrons. In plant seeds, there are cells specialized in storing energy as lipid droplets.
1. Morphology
Lipid droplets are observed as rounded and clear organelles located in the cytoplasm after general staining protocols (Figure 1). They are easily visualized at light microscopy after staining with liposoluble dyes such as Sudan black. Lipid droplets are variable in size and number depending on the cell type and physiological state of the cell. Many cells contain small lipid droplets, from 100 to 200 nm, whereas adipocytes of white fat may contain lipid droplets up to 200 µm in diameter. The number and size may change quickly in the same cell (Figure 2).
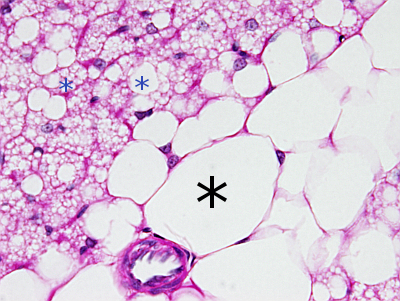

2. Functions
Lipid droplets perform many functions that may be common to all lipid droplets or depend on the cell type. Cell membrane lipids and cell energy may come from lipids stored in lipid droplets. Fat is a much better energy source than glycogen. Many enzymes involved in cellular fat metabolism are found on the surface of lipid droplets. A less known function is to prevent lipotoxicity by removing fatty acids from the bloodstream. High amount of circulating fatty acids may produce toxic molecules in the catabolic pathways. Lipids are also needed for synthesizing steroid hormones. In some animal species, adipocytes, with their large lipid droplets, form a thick layer under the skin that provides thermal insulation. In the cell, lipid droplets are associated with protein degradation centers and protect mitochondria and endoplasmic reticulum from oxidative stress.
Many proteins not related to the functions of the lipid droplet can be found on its surface. In this way, lipid droplets may function as transient protein stores. For instance, fruit fly embryos accumulate histones on the surface of lipid droplets, which are used later for the massive production of nuclei. Some viruses proteins concentrate on the lipid droplet surface, where the virus capsid is assembled.
In yeast, some lipid droplets remain physically connected to the endoplasmic reticulum. In mammals, lipid droplets are free in the cytoplasm, but lipid droplet-endoplasmic reticulum physical contacts are sometimes observed. These contacts make sense, since many enzymes synthesized in the endoplasmic reticulum are involved in the degradation and synthesis of fatty acids. In electron microscopy images, endoplasmic reticulum tubules are observed wrapping a lipid droplet, suggesting a strong communication between these two organelles. In addition, physical contacts have been observed between lipid droplets and mitochondria, lysosomes, peroxisomes, endosomes, and the nuclear envelope. Endosomes may also wrap lipid droplets, which may indicate a degradation pathway down to lysosomes by autophagy.
3. Composition
Lipid droplets are composed of neutral lipids surrounded by a membrane monolayer of amphipathic lipids with associated proteins. It is the only cell organelle delimited by a lipid monolayer. Lipid droplets contain neutral lipids, mostly triacylglycerols and cholesterol esters, in a variable proportion depending on the cell type. For instance, adipocytes have lipid droplets with a majority of triacylglycerols, whereas some macrophages show a higher proportion of cholesterol esters. The membrane monolayer largely consists of phospholipids (most are phosphatidylcholine, but also phosphatidylethanolamine and phosphatidylinositol), and cholesterol, with almost no sphingolipids. Many proteins with a variety of functions are inserted into this monolayer.
The proteins found in the lipid droplet monolayer may be synthesized in the endoplasmic reticulum or by free ribosomes in the cytosol. Inserting a protein into the monolayer is difficult because transmembrane proteins are intended for a membrane bilayer, and they do not fit comfortably in a monolayer. According to their origin, there are class I proteins coming from the endoplasmic reticulum and class II proteins coming from the cytosol (see below). Both show alpha chains of amino acids that can be accommodated in the membrane monolayer. How each protein is inserted is not clearly known yet.
In animal cells, perilipins are a family of proteins associated with lipid droplets. All lipid droplets in animal cells contain periliins. There are other proteins involved in lipid metabolism. About 50 types of proteins are estimated to be associated with lipid droplets. They can be classified according to their functions: structural, enzymes, involved in vesicular trafficking, signaling, and those that use the lipid droplet surface to carry out functions unrelated to the lipid droplet itself. Many of these proteins are also found in other cell compartments. For instance, triglyceride synthesizing enzymes are found in the endoplasmic reticulum membranes and on the surface of the lipid droplets. Other proteins may use the lipid droplets as a transient storage place, as mentioned above.
In plant cells, proteins associated with the lipid droplet surface are oleosins (structural proteins that can cross the membrane monolayer and are in contact with triglycerides), enzymes (caleosin, dioxygenase, stereoleosin, which are related to cell stress), and proteins associated with the hydrophobic segments of the membrane monolayer. The more abundant protein is oleosin, which may cover most of the lipid droplet surface, and contains an amino acid chain inserted among the triacylglycerols. The other proteins are usually attached to the lipid droplet surface and have a very short hydrophobic amino acid chain, or they do not have one at all.
4. Formation
Lipid droplets are initially formed by the accumulation of esterified lipids between the two hemilayers of the endoplasmic reticulum membrane (Figure 3). Resident proteins of the endoplasmic reticulum synthesize these lipids. For instance, DGAT1 and DGTA2 are involved in the synthesis of triacylglycerols. The lipid deposition in between both hemilayers forms a lens-like shape. This happens mostly in the endoplasmic reticulum tubules rather than in cisterns.
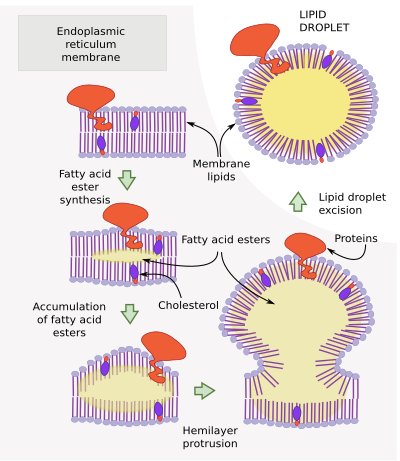
When a critical size is reached, the depot of lipids is released to the cytosol as a lipid droplet surrounded by a superficial hemilayer, which is the cytosolic hemilayer of the endoplasmic reticulum membrane. It is not known the critical size for the droplet scission and release, nor the proteins involved. However, the seipin proteins seem to indicate the point where the evagination is going to happen.
Once released, lipid droplets may grow due to the activity of lipid synthesizing enzymes coming from the endoplasmic reticulum. They are then referred to as expanding lipid droplets. They can also grow by fusion with other lipid droplets. Some new lipid droplets can be formed after the strangling of a large, already existing lipid droplet. The decrease in size and number of lipid droplets can be caused by two mechanisms: a higher activity of lipase enzymes found on the surface of the lipid droplet, or removal by autophagy.
Some proteins, like Pex 30, are found in the endoplasmic reticulum regions where lipid droplets are formed. Curiously, these proteins are also found in those regions of the endoplasmic reticulum that form peroxisomes. Thus, peroxisomes and lipid droplets may share a similar molecular machinery during their formation.
-
Bibliography ↷
-
Bibliography
Beller M, Thiel K, Thul PJ, Jäckle H. (2010).Lipid droplets: A dynamic organelle moves into focus. FEBS letters 584: 2176-2182.
Fujimoto T, Ohsaki Y. (2006). Annals of the academy of sciences of New York. 1086: 104-115.
Gao Q, Goodman JM. (2015).The lipid droplet—a well-connected organelle. Frotiers in cell and development biology.
Walther TC, Cheung J, Farese Jr, RV. (2017). Lipid droplets biogenesis. Annual review of cell and developmental biology. 33: 491-510.
Walther TC, Farese Jr.RV. (2012). Lipid droplets and cellular lipid metabolism. Annual review of biochemistry. 81: 687–714.
-
 Chloroplasts
Chloroplasts 