1. Structure
2. Organization
3. Miosin
3. Functions
- Cell shape
- Cell movement
- Internal organization
- Muscle contraction
- Cytokinesis
- Microvilli
Actin filaments are a component of the cytoskeleton. In animal cells, actin filaments are usually found near the plasma membrane (Figures 1 and 2), but their distribution and organization depend much on the cellular type. Actin filaments perform many functions in the cell, such as cell division, cell movement, endocytosis, phagocytosis, and organelle communication. In animal cells, actin filaments provide mechanical support for maintaining or changing the cellular shape.
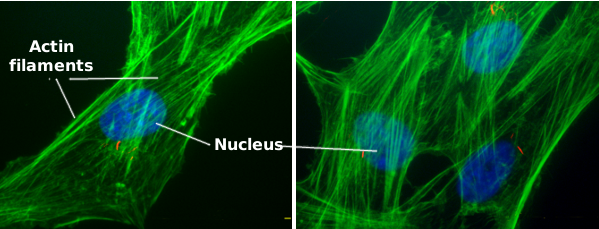
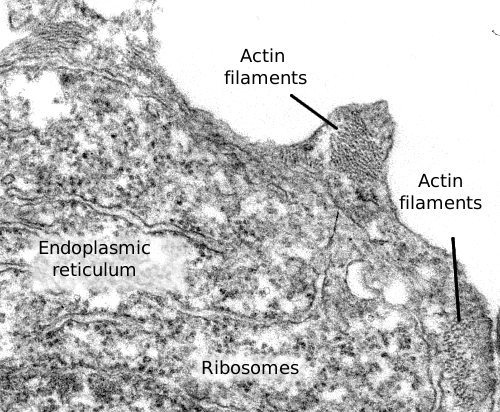
1. Structure
Actin filaments are built by polymerization of globular actin proteins (Figure 3), which can be found in two isoforms: alpha- and beta-actin. Alpha-actin is abundant in muscle cells. Beta-actin is the most frequent isoform, and it is found in most animal cells. Actin is a very abundant protein in the cytosol, accounting for about 10 % of the total cytosolic protein content. In the total cytosolic actin protein pool, some are found as part of the actin filaments (known as F-actin), and the remaining are free in the cytosol (known as G-actin).
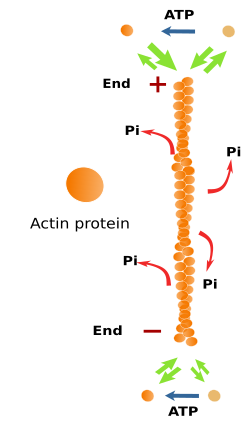
Actin filaments are 7 nm thick, thinner than the other cytoskeleton components (microtubules and intermediate filaments). That is why actin filaments are also known as microfilaments. Actin filaments have a minus end and a plus end, which means that they are polarized filaments. At the plus end, polymerization, the addition of new actin proteins, is more frequent than depolymerization, whereas at the minus end, depolymerization is more frequent. The increase and decrease in the microfilament length are caused by polymerization and depolymerization, respectively. In the cell, these changes are happening all the time. Actin filaments are the most dynamic components of the cytoskeleton.
The cytosolic environment and the concentration of free actin proteins in the cytosol prevent the spontaneous assembling of actin filaments. Hence, the formation of new microfilaments relies on nucleation protein complexes, such as Arp2/3 and formins. Arp2/3 proteins work as nucleation sites for new microfilaments, whereas formins stabilize transient spontaneous associations of actin proteins, boosting the formation and elongation rate of microfilaments. This control of microfilament formation is very useful for the cell because new microfilaments are formed when and where they are needed by precisely placing nucleation protein complexes.
2. Organization
A salient feature of actin filaments is that they are highly adaptable. They are formed and removed easily and are associated with each other in many ways to form 3D scaffolds. This versatility relies on more than 100 different modulator protein types, or actin associated proteins (Figure 4). These proteins regulate filament polymerization and depolymerization rates, the nucleation of new filaments, the destruction of existing filaments, and 3D organization. Actually, there are no naked actin filaments or free actin proteins in the cytosol, but they are always linked to some associated protein.
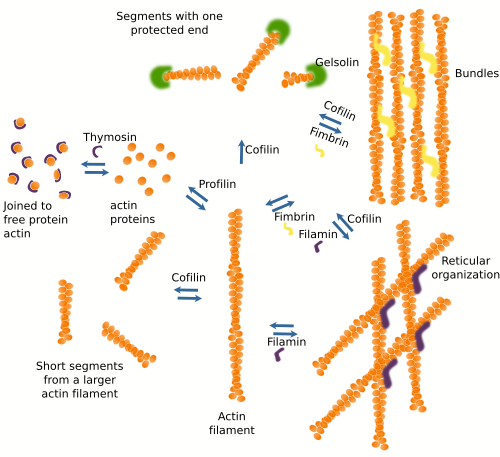
Proteins associated with actin filaments can be divided according to their functions. a) Modulate polymerization. Some proteins, such as profilin, join free actin and boost actin filament polymerization. Other proteins, such as thymosin, join free actin proteins and hamper the polymerization process by preventing the spontaneous polymerization of actin filaments. b) Modulate the spatial organization. There are proteins, such as fimbrin and alpha-actinin, that make cross-bridges between actin filaments to form bundles, whereas other proteins, like filamin, make the arrangement of actin filaments in reticular structures. c) Some actin binding proteins, such as cofilin, severin, and gelsolin, break and rearrange the actin filaments. d) There are also proteins that act as intermediates between actin filaments and other proteins. For example, tropomyosin mediates the interaction between actin filaments and myosin in the muscle cells. e) Some proteins are in charge of anchoring actin filaments to other cellular structures like membranes and other components of the cytoskeleton. Some actin associated proteins may perform more than one function.
There are additional elements modulating the effect of actin associated proteins on actin filament behavior, such as calcium concentration, activity of Rho-GTPases, presence of lipids, and higher or lower expression of their genes. There are also drugs that influence the polymerization of actin filaments. For example, cytochalasin inhibits polymerization, whereas phalloidin inhibits depolymerization.
3. Myosin
Many functions performed by actin filaments dependen on the type of motor proteins known as myosins. These proteins can move cargoes along the actin filament toward the plus end. The energy comes from ATP hydrolysis. Besides dragging cargoes, myosins can also slide two actin filaments, one over the other, in opposite directions. Furthermore, if the myosin is anchored, then the motor traction can move the actin filament. Myosins are actually a diverse family of proteins with more than 40 members in mammals.
4. Functions
Cell shape
Underneath the plasma membrane, there is a cortical layer of actin filaments about 100 nm thick (Figure 5). They are interlocked between each other by associated proteins and anchored to proteins and lipids of the plasma membrane. There are also myosins that generate mechanical forces that push actin filaments and change the position of the plasma membrane. This actin filament layer makes cells both withstand and generate mechanical forces, influencing, in this way, the cell shape. Animal cells lack a cell wall, so that cell shape largely depends on the actin filaments of the cortical cytoplasm.
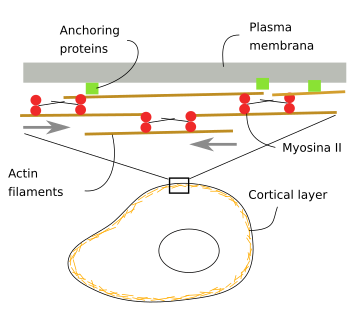
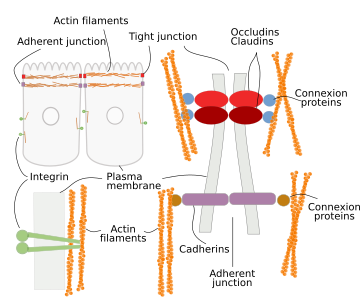
In animal cells, the cell shape also depends on the adhesion contacts with the extracellular matrix and other adjoining cells (Figure 6). Integrins mediate the adhesion of the cell to the extracellular matrix. In their cytosolic domain, integrins are connected to actin filaments, so there is a mechanical continuity between the extracellular matrix and cytoskeleton. Tight junctions and adherent junctions are cell-cell junctions made up of adhesion transmembrane proteins, occludin/claudin and cadherin, respectively, which are also connected to actin filaments by intermediary proteins.
Cell movement
Cells do not swim, but they crawl or trail across the tissues, as do, for example, the embryonic cells during development, amoebas when moving around, lymphocytes when heading toward damaged tissues, and the growth cone of neurons when searching for their targets. For cell movement, several steps are needed: the production and extension of cytoplasmic protrusions, the adhesion of these protrusions to some elements of the environment, and the drag the rest of the cell toward these anchoring points. The cellular protrusions are known as podia, and there are lamellipodia when they are sheet-like, filopodia when they are thin and long, and lobopodia when they are thick and tubular. Actin filaments have a prominent role in the formation of these protrusions (Figure 7). Actually, actin polymerization is what pushes the plasma membrane outward and forms protrusions.
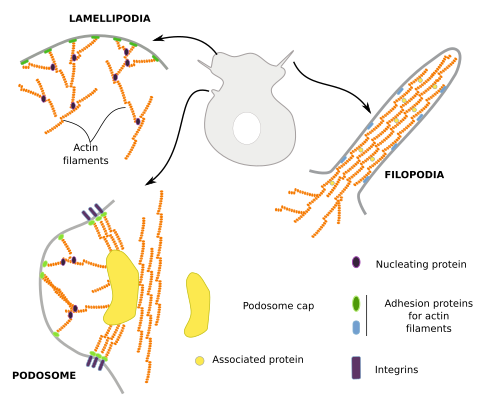
Lamellipodia are rather flat cell expansions originated by actin filament polymerization that, instead of bundles, form a branched scaffold. These expansions appear when cells are moving, and during phagocytosis and macropinocytosis. Filopodia may emerge from lamellipodia or from other cell regions. Typical filopodia are a few µm in diameter and no more than 10 µm in length. They contain a few dozens of actin filaments organized as a bundle. Podosomes are cell expansions attached to the extracellular matrix through integrins, but they are also able to release metalloproteinases for extracellular matrix degradation. They contain a central branched scaffold of actin filaments surrounded by non-branched actin filaments. Podosomes may be mechanosensors that explore the environment and may also contribute to cell movement.
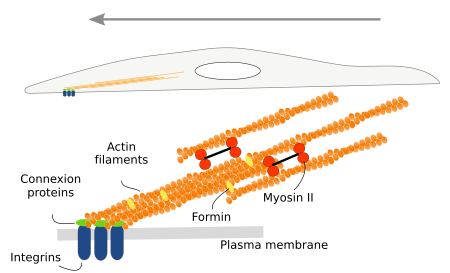
When these cell expansions contact any extracellular structure, either extracellular matrix macromolecules or other cells, they get anchored by adhesion proteins like integrins and cadherins. Once anchored, the cell pulls all the intracellular compartments and structures toward the adhesion points. The dragging force relies on the stress fibers, intracellular structures composed of actin filaments and myosin proteins (Figure 8).
Internal organization
Actin filaments are mostly found near the plasma membrane, in the so-called cortical region. In this place, they are involved in vesicle formation, macropinocytosis and phagocytosis (Figure 9).

Organelles are moved through the cytoplasm. Actin filaments, together with myosin motor proteins, help with the organelle movements (Figure 10). This role is very important in plant cells, where most of the internal movements of organelles are carried out by actin filaments. The chloroplast movement, a process known as cyclosis, can be observed under the light microscope.
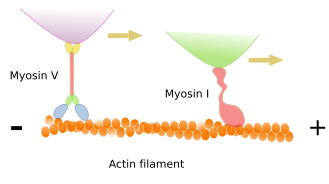
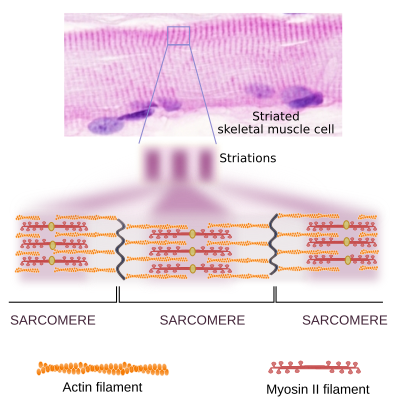
Muscle contraction
In muscle cells, myosin II molecules are associated with each other to form the thick myosin filaments, which show polarity as if they were an arrow with two arrowheads, each at one end (Figure 11). In the striated muscle, each of these heads drags actin filaments toward the middle point of the myosin filament. This is the molecular mechanism for muscle cell contraction.
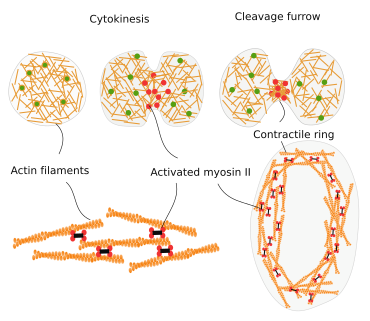
Cytokinesis
During cell division, the final strangling of the cytoplasm is produced by a ring of actin filaments that, helped by myosin II motor proteins, gets progressively smaller in diameter until the complete separation of the cytoplasm of the two new cells (Figure 12).
Microvilli
Microvilli are filiform expansions of the apical part of some cells that enormously increase the surface of the plasma membrane. For example, they can be found in the epithelium of the gut and in the proximal tube of the nephron. Each of these little expansions, or microvillus, is 1 to 2 µm height and around 0.1 µm thick. They contain several dozens of actin filaments oriented parallel to the longitudinal axis of the microvillus (Figure 13). At the base of microvilli, in the peripheral cytoplasm, there is a molecular scaffold known as the terminal web, which is composed of actin filaments and stabilizes the whole set of microvilli.

 Cytoskeleton
Cytoskeleton