Microvilli are short thin protrusions usually found in the free surfaces of some differentiated cells, more frequent in epithelial cells (Figures 1 and 2). They are filiform structures of about 1 to 2 µm long and around 100 nm thick. Inside, each microvillosity contains dozens of actin filaments oriented parallel to the long axis. Microvilli are usually dense and tightly packed like a brush border. In top view, microvilli are spatially organized as hexagons.
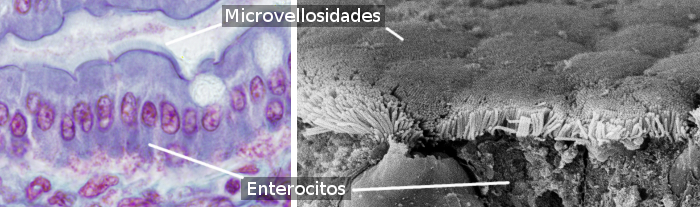
Figure 1. Small intestinal epithelium at light microscopy (image on the left) and at scanning electron microscopy (image on the right) showing microvilli at the free surface of the cells.
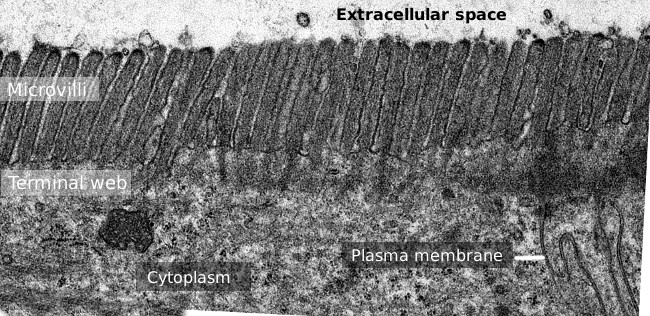
Figure 2. Transmission electron microscopy image of the small intestine epithelium surface. The terminal web of actin filaments is the darker band observed basal to microvilli.
There are many cell types bearing microvilli, but they are more frequently found in epithelial cells, like gut enterocytes, convoluted duct epithelium of kidneys and epididymus. Microvilli can also be found in some specialized sensory cells like olfactory receptor neurons, organ of Corti's receptor cells, placenta cells, and in cells which are moving. Although microvilli show similar external morphology in different cell types, they contain distinct molecular composition and show some differences in their internal structure. Below, we are dealing with the features of microvilli of enterocytes of the small intestine epithelium.
1. Formation
Microvilli are formed after actin filaments get associated with a dense plate found in the cytosolic surface of the plasma membrane. Proteins for gathering actin filaments in bundles and stabilizing them are also needed. It looks like it is a sequential process. First, villin facilitates the formation of bundles of actin filaments and stabilizes their distal ends inhibiting the filament grow, so villin may be important to establish the length of microvilli. Ezrin, less abundant, helps to connect the actin filaments of the bundle to the plasma membrane. A second phase of microvilli formation includes reaching the final length and the distribution on the cell surface as hexagons. Fimbrin and spin may contribute to these features. Lateral connections by protocadherins between actin filaments of different microvilli also happen during this second stage. It is important to notice that during microvilli formation a large amount of membrane and membrane proteins is needed, which is supplied by an intense exocytosis. Evaginations of plasma membrane, one per each microvillosity, is a consequence of actin filament polymerization.
2. Structure
Microvilli are made up of 6 main proteins: actin, fimbrin, villin, myosin (Myo1A), calmodulin and spectrin (non erythrocytic) (Figure 3). The microvellosity skeleton is a bundle of 30-40 actin filaments parallel to the long axis and with their minus ends toward the tip of the microvellosity. Fimbrin and villin link together the actin filaments, whereas Myo1A and calmodulin make the connections between the bundle and the membrane. The plus end of actin filaments extend into the cytoplasm and there the actin filaments of different microvilli intermingle between each other and form a web with a hexagonal pattern. This web is known as terminal web, and it spreads through the cell periphery under the microvilli. Terminal web contains large amount of non-erythrocytic spectrin.
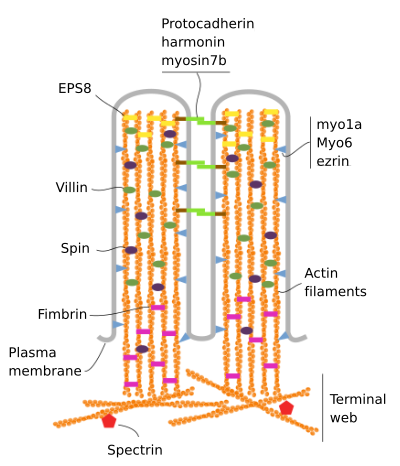
Figure 3. Molecular organization of microvilli (adapted from Crawley et al., 2014)
Each microvellosity is stable and does not move, but its protein skeleton is in permanent renewing by exchanging actin and other scaffold proteins between the microvellosity and the cytosol. It means that there is a balance between incoming and outgoing proteins. It is estimated that every 20 minutes the skeleton of each microvellosity is completed renewed. Microvilli may be completely disorganized and disappear after an increase in calcium concentration, for example under cell stress. Calcium changes the behavior of villin, from stabilizing to severing actin filaments. Microvilli disappear in cells entering mitosis. The terminal web is also a plastic structure.
3. Function
Exchange of substances between cavities or external environment and tissues is one the main function of epithelia, such as intestine and kidney epithelia. It is mostly done at the apical (free) surface of the epithelial cells, where most molecular transporter, ion pumps are located, and endocytic processes happen. The more cell surface the more space to accommodate the molecular machinery for the exchange. Microvilli are spine-like protrusions that increase enormously the plasma membrane surface and therefore the membrane protein content. This particularly important in those absorptive and secretory cells of epithelia. Microvilli may increase the cell surface up to 100 times compared with a flat membrane. Enterocyte microvilli bear many enzymes, which are not involved in molecular exchange, but in digestion.
Microvilli may influence transduction molecular pathways. Their membranes work as relatively isolated domains with a distinct set of molecules, like glucose transporters, ion pumps and receptors. The length of microvilli is well suited for performing local transduction of the molecular pathways independently of the cytosol. In addition, the actin filaments and structural proteins scaffold form a dense network of molecules that somehow filters or controls the molecules coming or quitting the interior of each microvellosity. This scaffold also may work as a temporal storage for calcium.
The high density of microvilli makes a physical barrier that protects against pathogens. This is important for intestine epithelium. In addition, the large amount of membrane stored in microvilli is a reservoir that can withstand hypertonic insults and prevent the breakage of the plasma membrane.
Stereocilia are specialized microvillosities performing sensory functions. Despite the name stereocilium, they are actually modified microvillosities transformed into sensory structures, and some authors propose the name estereomicrovellosity instead. They can be found in the epididymis epithelium, and in the inner ear, working as mechanoreceptors that sense the surrounding fluid movement. Sterocilia of the mammal inner ear are found as part of the Corti's organ. They are 10 to 50 µm in length and contain more than 3000 actin filament inside. The sound waves of the air are transformed in fluid waves in the ear, which stereocilia sense and transform into electrical signals that travels via auditory nerve to the brain. Stereocilia are like antennas that receive external signals, which are transduced in electrical information.
Some microvellosities are specialized in sensing light. Photorreceptors are light sensory cells found in visual organs like the retina. The cellular sensory structures are actually modified cilia or microvellosities. Photoreceptors bearing modified microvellosities for light detection can be found in invertebrates visual structures. Those photoreceptors contain microvellosities arranged in the so-called rhabdomeres. Each microvellosity contains visual pigments that can respond to low light intensities and are efficient at high light intensities. Microvellosities organize forming the so-called rhabdomere, and the transduction molecular pathway make microvillosity based light response more sensible than cilium light sensory systems of vertebrates.
A large number of extracellular vesicles are proposed to be released from microvilli. The tips of enterocyte microvilli are able to release vesicles to the surrounding environment. The connection between plasma membrane and actin-myosin network may help to produce these vesicles by dragging membrane toward the distal part of each microvellosity, where small portions of membrane are transformed in vesicles. These extracellular vesicles contain enzymes and are enriched in alkaline phosphatase.
Bibliography
Brown J W, McKnight C J. 2010. Molecular model of the microvillar cytoskeleton and organization of the brush border. PLoS One. 5: e940 .
Crawley SW, Mooseker MS, Tyska MJ. 2014. Shaping the intestinal brush border. J Cell Biol. 207: 441-451 .
Fain G L, Hardie R, Laughlin S B. 2010. Phototransduction and the evolution of photoreceptors. Curr Biol. 20: R114-R124 .
Lange K. 2011. Fundamental role of microvilli in the main functions of differentiated cells: Outline of an universal regulating and signaling system at the cell periphery. J Cell Physiol. 226: 896-92
McConnell R E, Higginbotham J N, Shifrin Jr D A, Tabb D L, Coffey R J, Tyska M J. 2009. The enterocyte microvillus is a vesicle-generating organelle. J Cell Biol. 185: 1285-129 .
Sauvanet C, Wayt J, Pelaseyed T, Bretscher A. 2015. Structure, regulation, and functional diversity of microvilli on the apical domain of epithelial cells. Annual review of cell and development biology. 31: 593-621.